Advertisements
Advertisements
Question
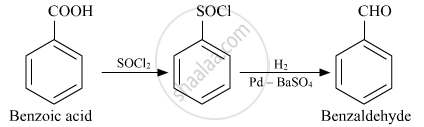
How will you convert the following in not more than two steps:
Benzoic acid to Benzaldehyde
Advertisements
Solution
Benzoic acid to Benzaldehyde
APPEARS IN
RELATED QUESTIONS
Derive van’t Hoff general solution equation.
19.5 g of CH2FCOOH is dissolved in 500 g of water. The depression in the freezing point of water observed is 1.0°C. Calculate the van’t Hoff factor and dissociation constant of fluoroacetic acid.
How van’t Hoff factor is related to the degree of dissociation?
How will you convert the following in not more than two steps:
Acetophenone to Benzoic acid
Give reasons for the following
Elevation of the boiling point of 1 M KCl solution is nearly double than that of 1 M sugar solution.
Predict whether van’t Hoff factor, (i) is less than one or greater than one in the following:
CH3COOH dissolved in water
Phenol dimerizes in benzene having van’t Hoff factor 0.54. What is the degree of association?
Maximum lowering of vapour pressure is observed in the case of ______.
Consider the reaction
\[\begin{bmatrix}\begin{array}{cc}
\phantom{.......}\ce{CH3}\\
\phantom{....}|\\
\ce{CH3CH2CH2 - \overset{⊕}{N} - CH2CH3}\\
\phantom{....}|\\
\phantom{.......}\ce{CH3}
\end{array}\end{bmatrix}\]\[\ce{OH^- ->[Heat] ?}\]
Which of the following is formed in a major amount?
Why is boiling point of 1 M NaCl solution more than that of 1 M glucose solution?
