Advertisements
Advertisements
प्रश्न
How will you convert the following in not more than two steps:
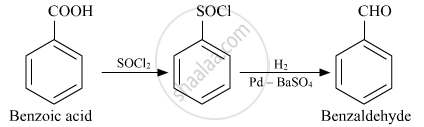
Benzoic acid to Benzaldehyde
Advertisements
उत्तर
Benzoic acid to Benzaldehyde
APPEARS IN
संबंधित प्रश्न
Derive van’t Hoff general solution equation.
Calculate the amount of benzoic acid (C6H5COOH) required for preparing 250 mL of 0.15 M solution in methanol.
Give reasons for the following
Elevation of the boiling point of 1 M KCl solution is nearly double than that of 1 M sugar solution.
Predict whether van’t Hoff factor, (i) is less than one or greater than one in the following:
CH3COOH dissolved in water
The Van't Hoff factor (i) for a dilute aqueous solution of the strong elecrolyte barium hydroxide is (NEET) ______.
Phenol dimerizes in benzene having van’t Hoff factor 0.54. What is the degree of association?
Van't Hoff factor I is given by expression.
What is the expected each water van't Hoff factor for and K4[F4(CN6)] when it completely dissociated in waters.
Why is boiling point of 1 M NaCl solution more than that of 1 M glucose solution?
Calculate Van't Hoff factor for an aqueous solution of K3 [Fe(CN)6] if the degree of dissociation (α) is 0.852. What will be boiling point of this solution if its concentration is 1 molal? (Kb = 0.52 K kg/mol)
