Advertisements
Advertisements
प्रश्न
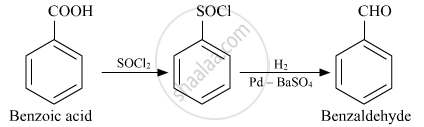
How will you convert the following in not more than two steps:
Benzoic acid to Benzaldehyde
Advertisements
उत्तर
Benzoic acid to Benzaldehyde
APPEARS IN
संबंधित प्रश्न
19.5 g of CH2FCOOH is dissolved in 500 g of water. The depression in the freezing point of water observed is 1.0°C. Calculate the van’t Hoff factor and dissociation constant of fluoroacetic acid.
Define the term abnormal molar mass.
How van’t Hoff factor is related to the degree of dissociation?
Give reasons for the following
Elevation of the boiling point of 1 M KCl solution is nearly double than that of 1 M sugar solution.
Predict whether van’t Hoff factor, (i) is less than one or greater than one in the following:
CH3COOH dissolved in water
Van’t Hoff factor i is given by the expression:
(i) i = `"Normal molar mass"/"Abnormal molar mass"`
(ii) i = `"Abnormal molar mass"/"Normal molar mass"`
(iii) i = `"Observed colligative property"/"Calculated colligative property"`
(iv) i = `"Calculated colligative property"/"Observed colligative property"`
Maximum lowering of vapour pressure is observed in the case of ______.
Geraniol, a volatile organic compound, is a component of rose oil. The density of the vapour is 0.46 g L–1 at 257°C and 100 mm Hg. The molar mass of geraniol is ______ g mol–1. (Nearest Integer)
[Given: R = 0.082 L atm K–1 mol–1]
When 9.45 g of ClCH2COOH is added to 500 mL of water, its freezing point drops by 0.5°C. The dissociation constant of ClCH2COOH is x × 10−3. The value of x is ______. (Rounded-off to the nearest integer)
[\[\ce{K_{f(H_2O)}}\] = 1.86 K kg mol−1]
A molecule M associates in a given solvent according to the equation \[\ce{M <=> (M)_n}\]. For a certain concentration of M, the van't Hoff factor was found to be 0.9 and the fraction of associated molecules was 0.2. The value of n is ______.
