Advertisements
Advertisements
Question
Give the main experimental points only to demonstrate that candle-gains weight on burning.
Advertisements
Solution
Candle gains weight on burning
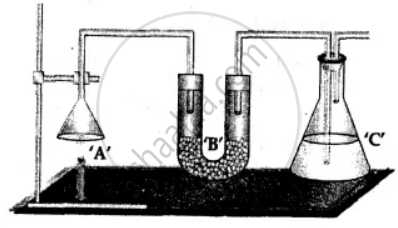
1. Weight: The complete apparatus as shown above which includes -
- Unlit candle ‘A’.
- U tube ‘B’ containing anhydrous CaCl2.
- Conical flask ‘C’ containing cone. KOH soln.
Total wt. = a gms.
The candle ‘A’ is then lit and the products obtained on burning are allowed to be absorbed in U tube ‘B’ and conical flask ‘C’.
2. Re-weigh: The complete apparatus as shown above after the candle has burnt – for a known period of time.
Total wt. = b gms.
Result:
- ‘b’ gms. is greater than ‘a’ gms.
- weight of apparatus after absorbtion
- of products is greater than the original weight of the apparatus.
Conclusion: Candle gains weight due to mass of oxygen of the air which has combined with ‘carbon’ and ‘hydrogen’ of the candle [CxHy] producing .
- Water vapour [absorbed by anhydrous CaCl2]
- Carbon dioxide [absorbed by cone. KOH soln].
APPEARS IN
RELATED QUESTIONS
Define the term acid rain.
A metal highly resistant to rusting.
State two differences between:
Rusting and burning.
The process by which oxidation of food in our body takes place is
What is rusting?
State how nitrogen is utilised by plants for their growth. Give a reason why nitrogen is used in flushing food packages.
Describe simple experiments to show the presence of -
oxygen and nitrogen component in air using a bell jar
Describe simple experiments to show the presence of -
water vapour in air using a glass tumbler and ice.
In the laboratory preparation of oxygen from hydrogen peroxide answer the following:
Name the catalyst used in the preparation and state it’s function.
A process without emitting flame is called as ______.
