Advertisements
Advertisements
प्रश्न
Give the main experimental points only to demonstrate that candle-gains weight on burning.
Advertisements
उत्तर
Candle gains weight on burning
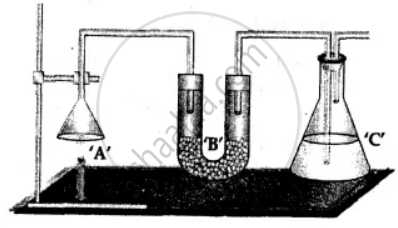
1. Weight: The complete apparatus as shown above which includes -
- Unlit candle ‘A’.
- U tube ‘B’ containing anhydrous CaCl2.
- Conical flask ‘C’ containing cone. KOH soln.
Total wt. = a gms.
The candle ‘A’ is then lit and the products obtained on burning are allowed to be absorbed in U tube ‘B’ and conical flask ‘C’.
2. Re-weigh: The complete apparatus as shown above after the candle has burnt – for a known period of time.
Total wt. = b gms.
Result:
- ‘b’ gms. is greater than ‘a’ gms.
- weight of apparatus after absorbtion
- of products is greater than the original weight of the apparatus.
Conclusion: Candle gains weight due to mass of oxygen of the air which has combined with ‘carbon’ and ‘hydrogen’ of the candle [CxHy] producing .
- Water vapour [absorbed by anhydrous CaCl2]
- Carbon dioxide [absorbed by cone. KOH soln].
APPEARS IN
संबंधित प्रश्न
State two differences between:
Rusting and burning.
Fill in the blanks :
Oxides of sulphur and nitrogen combine with rain water to form ___________ and _________ which cause ____________.
The process by which oxidation of food in our body takes place is
What happens when potassium nitrate is heated ?
Tick (√ ) the most appropriate answer.
Galvanising is the process of coating iron with a layer of molten :
How will you prove that oxide formed by burning sodium is basic in nature?
State four ways of preventing rusting.
State how nitrogen is utilised by plants for their growth. Give a reason why nitrogen is used in flushing food packages.
Describe simple experiments to show the presence of -
carbon dioxide in air using a test tube with outlets containing lime water.
Name the following:
The pollutant which combines with water vapour to give sulphuric acid – a product of acid rain.
