Advertisements
Advertisements
Question
Give the main experimental points only to demonstrate that magnesium.
Advertisements
Solution
Experiment to show that magnesium gains weight on burning:
Take dry crucible with lid. Put a small piece of magnesium in it and cover the lid. Find its weight with lid covering it. Let it be 50 gms.
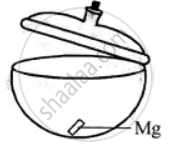
Now heat the crucible along with magnesium and lid just opened for a while. Taking care that no ash comes out. Let it cool covering the lid and products formed weight it again. The weight now will be more. Let it be 50.82 gms, oxygen combined is 50.82 – 50 = 0.82 gms this shows that something is added to magnesium, which has increased its weight. This 0.82 gm is the weight of oxygen added to magnesium.
2Mg + O2 → 2MgO
Hence magnesium gains weight on burning.
APPEARS IN
RELATED QUESTIONS
Define the term acid rain.
A mixture of oxygen and carbon dioxide used for artificial respiration.
State and explain two ways by which air gets polluted in nature.
Tick (√ ) the most appropriate answer.
Galvanising is the process of coating iron with a layer of molten :
How will you prove that oxide formed by burning sodium is basic in nature?
Describe simple experiments to show the presence of -
water vapour in air using a glass tumbler and ice.
In the laboratory preparation of oxygen from hydrogen peroxide answer the following:
Name the catalyst used in the preparation and state it’s function.
Distinguish between:
Combustion and Respiration
In Rockets, along with fuel, oxygen is also carried for combustion – why?
A process without emitting flame is called as ______.
