Advertisements
Advertisements
Question
Describe simple experiments to show the presence of -
oxygen and nitrogen component in air using a bell jar
Advertisements
Solution
Oxygen and nitrogen in air:
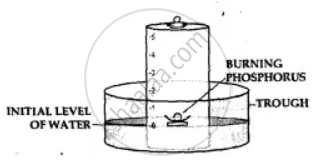
Apparatus— A through, a bell jar, a cork and a crucible and a piece of white phosphorus.
producer-
- The trough is filled with water and a bell jar divided into 5 equal parts, ‘1 to 5’ is placed over it.
- A crucible containing white phosphorus is placed on a cork which is made to float on water.
- The level of the water inside and outside are adjusted to one level.
- The phosphorus is then ignited by means of a heated wire.
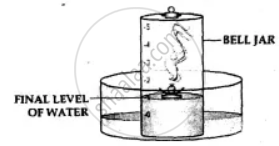
Observation-
- The phosphorus burns in the active component of air [oxygen] forming – dense white fumes of phosphorus pentoxide [P2O5]. Phosphorus + Oxygen →Phosphorus pentoxide
- The level of the water in the bell jar rises by l/5th.
Conclusion-
- Oxygen— The active component of air, i.e. 1/5th of air, is used up in burning.
- Nitrogen— The inactive component of air, i.e. 4/ 5 of air, is not used up in burning. Hence air contains oxygen which supports combustion and nitrogen which does not.
APPEARS IN
RELATED QUESTIONS
Define the term acid rain.
State two differences between:
Rusting and burning.
Fill in the blanks :
Oxides of sulphur and nitrogen combine with rain water to form ___________ and _________ which cause ____________.
The process by which oxidation of food in our body takes place is
What happens when potassium nitrate is heated ?
How will you prove that oxide formed by burning sodium is basic in nature?
State two most important conditions for rusting.
Write the balanced chemical equation of the manganese dioxide.
Distinguish between:
Combustion & Rusting with suitable word equations only?
Match the following
| 1. | Burning of meteors | a. | Exosphere |
| 2. | Weather change | b. | Stratosphere |
| 3. | Lowest temperature | c. | Mesosphere |
| 4. | Ozone layer | d. | Troposphere |
