Advertisements
Advertisements
प्रश्न
Describe simple experiments to show the presence of -
oxygen and nitrogen component in air using a bell jar
Advertisements
उत्तर
Oxygen and nitrogen in air:
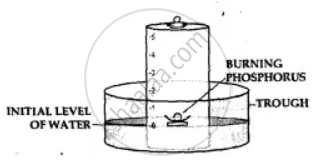
Apparatus— A through, a bell jar, a cork and a crucible and a piece of white phosphorus.
producer-
- The trough is filled with water and a bell jar divided into 5 equal parts, ‘1 to 5’ is placed over it.
- A crucible containing white phosphorus is placed on a cork which is made to float on water.
- The level of the water inside and outside are adjusted to one level.
- The phosphorus is then ignited by means of a heated wire.
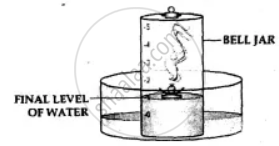
Observation-
- The phosphorus burns in the active component of air [oxygen] forming – dense white fumes of phosphorus pentoxide [P2O5]. Phosphorus + Oxygen →Phosphorus pentoxide
- The level of the water in the bell jar rises by l/5th.
Conclusion-
- Oxygen— The active component of air, i.e. 1/5th of air, is used up in burning.
- Nitrogen— The inactive component of air, i.e. 4/ 5 of air, is not used up in burning. Hence air contains oxygen which supports combustion and nitrogen which does not.
APPEARS IN
संबंधित प्रश्न
What do you observe when Magnesium is heated and then tested with moist blue and red litmus – paper?
State two differences between:
Rusting and burning.
State and explain two ways by which air gets polluted in nature.
Fill in the blank space by choosing the correct word from the given list.
List: sulphurous, nitric acid, red lead oxide paint, oxygen, phosphoric
To prevent rusting, the underside of the ships is coated with __________________.
State four ways of preventing rusting.
Write the balanced chemical equation of the manganese dioxide.
Identify the acidic oxides responsible for acid rain. State how their presence results in formation of acid rain. Give a reason why acid rain damages heritage buildings.
In the laboratory preparation of oxygen from hydrogen peroxide answer the following:
Draw a neat labelled diagram for the method used in the above preparation.
Give the main experimental points only to demonstrate that candle-gains weight on burning.
Give reason for the following:
Combustion and respiration show similarity.
