Advertisements
Advertisements
Question
Give the general characteristics of organic compounds.
Advertisements
Solution
All organic compounds have the following characteristic properties.
- They are covalent compounds of carbon and generally insoluble in water and readily soluble in organic solvent such as benzene, toluene, ether, chloroform etc…
- Many of the organic compounds are inflammable (except CCl4). They possess low boiling and melting points due to their covalent nature.
- Organic compounds are characterized by functional groups. A functional group is an atom or a specific combination of bonded atoms that react in a characteristic way, irrespective of the organic molecule in which it is present. In almost all cases, the reaction of an organic compound takes place at the functional group. They exhibit isomerism which is a unique phenomenon.
APPEARS IN
RELATED QUESTIONS
Find out all the functional groups present in the following polyfunctional compound.
Thyroxine, the principal thyroid hormone.

Identify primary, secondary, tertiary and quaternary carbon in the following compound.
How many cyclic and acyclic isomers are possible for the molecular formula C3H6O?
Identify the pair of organic compounds which belongs to homologous series.
An organic compound containing carbon, hydrogen and oxygen is soluble in dil. H2SO4 and does not react with sodium metal or KMnO4. When it is heated with HI in excess, a single alkyl halide is obtained. The original compound can be ____________.
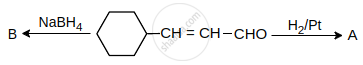
A and B are:
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{.....}|\phantom{...............}\\
\ce{CH3}\phantom{..}\ce{CH3}\phantom{.............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}\\
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{.....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
