Advertisements
Advertisements
Question
Explain with the help of a balanced equation the brown ring test for nitric acid.
Advertisements
Solution
Procedure:
- Add freshly prepared saturated solution of iron (II) sulphate to the aq. solution of nitric acid.
- Now add conc. sulphuric acid carefully from the sides of the test tube so that it does not fall dropwise in the test tube.
- Cool the test tube in water.
- A brown ring appears at the junction of the two liquids.
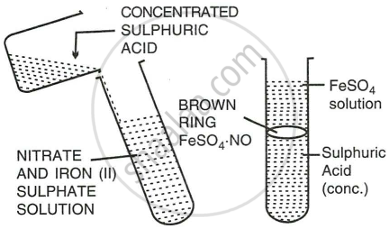
Reaction:
\[\ce{6FeSO4 + 3H2SO4 + 2HNO3 -> 3Fe2(SO4)3 + 4H2O + 2NO}\]
\[\ce{FeSO4 + NO -> \underset{a brown compound]}{\underset{[Nitroso ferrous sulphate,}{FeSO4*NO}}}\]
APPEARS IN
RELATED QUESTIONS
Name the products formed when dilute HNO3 is added to copper.
Give two chemical equations for the following:
Nitric acid acting as the oxidizing agent.
Write an equation for the following conversions A, B, C and D.

How will you prepare the following from nitric acid?
Aqua regia
What is the purpose of Conc. H2SO4 in the above preparation?
Nitric acid cannot be concentrated beyond 68% by the distillation of a dilute solution of HNO3, State the reason.
Give reason for the following:
Nitric acid is used in purification of gold.
What do you observe when?
Scrap zinc is heated with conc. HNO3?
What happen when (Given balanced equation):
Limestone reacts with nitric acid?
Balanced equation of oxidation of carbon with concentrated HNO3
