Advertisements
Advertisements
प्रश्न
Explain with the help of a balanced equation the brown ring test for nitric acid.
Advertisements
उत्तर
Procedure:
- Add freshly prepared saturated solution of iron (II) sulphate to the aq. solution of nitric acid.
- Now add conc. sulphuric acid carefully from the sides of the test tube so that it does not fall dropwise in the test tube.
- Cool the test tube in water.
- A brown ring appears at the junction of the two liquids.
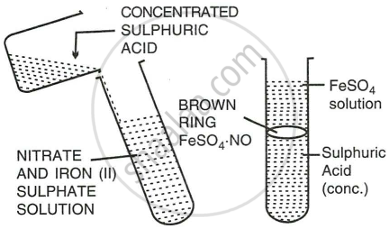
Reaction:
\[\ce{6FeSO4 + 3H2SO4 + 2HNO3 -> 3Fe2(SO4)3 + 4H2O + 2NO}\]
\[\ce{FeSO4 + NO -> \underset{a brown compound]}{\underset{[Nitroso ferrous sulphate,}{FeSO4*NO}}}\]
APPEARS IN
संबंधित प्रश्न
Give a balanced chemical equation for Laboratory preparation of Nitric acid.
Explain the following:
An all-glass apparatus is used in the laboratory preparation of nitric acid.
Give two chemical equations for the following:
Reactions of nitric acid with non-metals.
How will you prepare the following from nitric acid?
Aqua regia
Name the following :
Gas produced in air during lighting.
Give two chemical tests for nitric acid.
Write balanced equation and name the product formed when :
Cupric oxide reacts with nitric acid.
Write balanced equation for the reaction between nitrogen and oxygen, when lightning strikes.
Lead nitrate decomposes on heating to give ______.
The figure given below illustrates the apparatus used in the laboratory preparation of nitric acid.

- Name A (a liquid), B (a solid), and C (a liquid). (Do not give the formulae.)
- Write an equation to show how nitric acid undergoes decomposition.
- Write the equation for the reaction in which copper is oxidised by concentrated nitric acid.
