Advertisements
Advertisements
प्रश्न
Explain with the help of a balanced equation the brown ring test for nitric acid.
Advertisements
उत्तर
Procedure:
- Add freshly prepared saturated solution of iron (II) sulphate to the aq. solution of nitric acid.
- Now add conc. sulphuric acid carefully from the sides of the test tube so that it does not fall dropwise in the test tube.
- Cool the test tube in water.
- A brown ring appears at the junction of the two liquids.
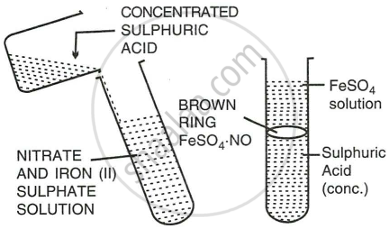
Reaction:
\[\ce{6FeSO4 + 3H2SO4 + 2HNO3 -> 3Fe2(SO4)3 + 4H2O + 2NO}\]
\[\ce{FeSO4 + NO -> \underset{a brown compound]}{\underset{[Nitroso ferrous sulphate,}{FeSO4*NO}}}\]
APPEARS IN
संबंधित प्रश्न
What is the type of salt formed when the reactants are heated at a suitable temperature for the preparation of Nitric acid?
Name a metal nitrate which on heating is changed into metal oxide.
Name the catalyst used in Ostwald process.
Explain with the help of a balanced equation, the brown ring test for nitric acid.
What do you observe when?
Conc. HNO3 drops on the skin of a person?
What happens when (Given balanced equation) :
Sulphar is added to hot and conc. HNO3?
What happen when (Given balanced equation):
Nitric acid is added to washing soda and the resulting gas is passed through a freshly prepared lime water?
Write the equation for the reaction of dilute nitric acid with copper.
Complete the table:
| Name of Process | Inputs | Equation | Output |
| Ammonia + Air | Nitric acid |
Give a balanced equation for the following reaction:
Copper reacts with concentrated nitric acid.
