Advertisements
Advertisements
Question
Explain the following:
Lead carbonate does not react with dilute HCl.
Advertisements
Solution
The salt produced is insoluble in the solution so the reaction does not proceed. Hence, we do not expect lead carbonate to react with hydrochloric acid.
APPEARS IN
RELATED QUESTIONS
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with zinc granules.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with iron filings.
What ions are present in the solutions of following substances? (write the symbols only)
Hydrochloric acid
Write the chemical formula of soda ash?
If you take some distilled water in a test-tube, add an equal amount of acetic acid to it, shake the test-tube well and leave it undisturbed on the test-tube stand, then after about 5 minutes what would you observe?
(A) There is a layer of water over the layer of acetic acid.
(B) A precipitate is setting at the bottom of the test-tube.
(C) Bubbles of colourless gas are coming out of the test-tube.
(D) There is a clear, colourless transparent solutions in the test-tube.
Magnesium hydroxides are used for treating _______
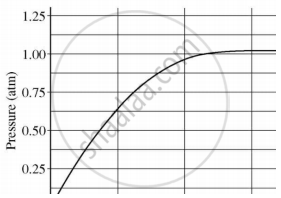
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
Take a clean test tube with a holder and pour some dilute hydrochloric acid. Add a few pieces of magnesium ribbon pieces slowly. What do you observe? Now show a burning match stick near the mouth of the test tube. Do you hear any sound? The gas burns with a pop sound. From this, it is observed that hydrogen gas has been formed due to the reaction between acid and metal.
“All acids contain one or more hydrogen atoms, but not all substances containing hydrogen are acids.” Support this statement with examples.
Consider the following salt:
YCl
What would be the pH of the solution if, in YCl, Y is sodium? Give a reason for your answer.
