Advertisements
Advertisements
Question
Explain in brief the experimental proof which led to the discovery of –
Atomic nucleus
Advertisements
Solution
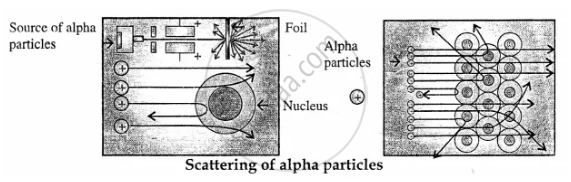
Discovery of Atomic Nucleus: Lord Rutherford in 1911 directed alpha particles (42He) towards gold foil. The deflection of the alpha particles was observed and concluded that there was a nucleus at the centre of atom which was positively charged dense very small space (solid) called the nucleus.
APPEARS IN
RELATED QUESTIONS
Multiple Choice Questions
The number of electrons in an atom is equal to number of
Complete the table below by identifying A, B, C, D, E and F.
FILL IN THE BLANK
Nuclear energy can be used to make ..........................
Tick the most appropriate answer.
Which of the following is used as a coolant in nuclear reactor?
- liquid sodium
- liquid nitrogen
- cadmium steel rods
- plutonium
TRUE \ FALSE
Uranium-235 and uranium-238 both undergo nuclear fission.
WRITE SHORT ANSWER
What is the central part of an atom known as?
State one major drawback of Rutherford's model.
Name the following:
The element which does not contain any neutron in its nucleus.
Explain in brief – Lod Rutherford’s experiment which led to the discovery of the atomic nucleus.
‘Electrons revolve around the nucleus in fixed orbits or shells called energy levels’. State how these energy levels are represented.
