Advertisements
Advertisements
प्रश्न
Explain in brief the experimental proof which led to the discovery of –
Atomic nucleus
Advertisements
उत्तर
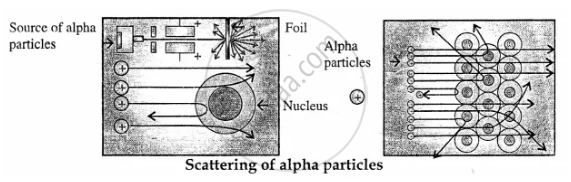
Discovery of Atomic Nucleus: Lord Rutherford in 1911 directed alpha particles (42He) towards gold foil. The deflection of the alpha particles was observed and concluded that there was a nucleus at the centre of atom which was positively charged dense very small space (solid) called the nucleus.
APPEARS IN
संबंधित प्रश्न
Give the following a suitable word/phrase.
The number of protons present in the nucleus of an atom.
How does the existence of isotopes contradict Dalton’s atomic theory?
FILL IN THE BLANK
Amount of energy stored inside the ..................... is known as nuclear energy.
Tick the most appropriate answer.
Which of the following is used as a coolant in nuclear reactor?
- liquid sodium
- liquid nitrogen
- cadmium steel rods
- plutonium
TRUE \ FALSE
Uranium-235 and uranium-238 both undergo nuclear fission.
ANSWER IN DETAIL
What are the various components of a nuclear power plant?
Which part of atom was discovered by Rutherford?
Which metal did rutherford select for his `alpha` particle scattering experiment and why?
‘Electrons revolve around the nucleus in fixed orbits or shells called energy levels’. State how these energy levels are represented.
What is meant by the term nucleus
