Advertisements
Advertisements
Question
Explain effects of lanthanoid contraction.
Advertisements
Solution 1
- Effects on the basic strength of hydroxides: As the size of the lanthanoid ions decreases from La3+ to Lu3+, the covalent character of the hydroxides increases. Thus, the basic strength decreases. Hence, La(OH)3 is most basic, while Lu(OH)3 is the least basic.
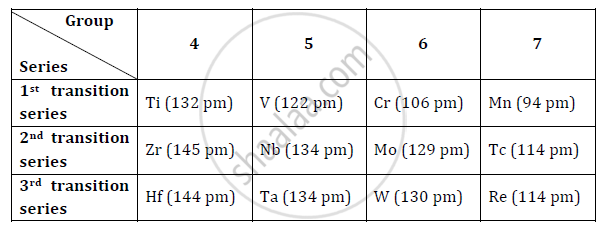
- Ionic radii of post lanthanoids: The elements which follow the lanthanoids in the third transition series are known as post lanthanoids. As a result of lanthanoid contraction, the atomic radii (size) of the elements which follow lanthanum (Hf, Ta, W etc.) are similar to those of the elements of the previous period. There is normal increase in size from Sc to Y to La. This trend disappears after the lanthanoids, and pairs of elements Zr–Hf (group 4), Nb–Ta (group 5), Mo–W (group 6) and Tc–Re (group 7) have almost identical sizes. These atoms possess similar number of valence electrons and similar properties. These pairs of elements are called ‘chemical twins’. The elements of the second and third transition series resemble each other more closely than the elements of the first and second transition series.
- Difficulty in separation of lanthanoids:- Because the changes in ionic radii (size of the ions) in the lanthanoids are very small, their chemical properties are similar. This makes the separation of the lanthanoids in the pure state difficult. However, lanthanoid contraction results in slight difference in the size of the lanthanoids which results in the differences in properties such as solubility, complex formation, hydration, basic character of their hydroxides etc. Because of these differences, the lanthanoids can be separated by the ion exchange method.
Solution 2
I. Decrease in Basicity:
- As per Fajan’s principle, “With increase in the size of cation, the tendency of the hydroxide to dissociate, increases. This increases the strength of base.”
- The size of the Ln3+ cation decreases with increase in the atomic number, due
to lanthanoid contraction. - This decreases the ionic character of M−OH bond and gradually increases the covalent character of M−OH bond.
- Therefore, the basic strength of the corresponding hydroxides decreases
from La(OH)3 to Lu(OH)3. - Thus, La(OH)3 is most basic and Lu(OH)3 is least basic.
II. Ionic radii of post-lanthanoids:
- The elements which follow the lanthanoids in the third transition series are known as post-lanthanoids.
- There is a regular increase in size from Sc to Y to La.
- But after the lanthanoids, the increase in radii from second to third transition series almost vanishes.
- Pairs of elements such as Zr–Hf (group 4), Nb–Ta (group 5), Mo–W (group 6) and Tc-Re (group 7) possess almost same size. These pair of elements are called ‘chemical twins’. The properties of these elements are also similar. So, due to lanthanoid contraction, elements of the second and third series resemble each other.
APPEARS IN
RELATED QUESTIONS
Which among the following pairs of elements is 'not' an example of chemical twins?
Account for the following :
Zr and Hf have almost similar atomic radii.
What are the different oxidation states exhibited by the lanthanoids?
Compare the chemistry of actinoids with that of the lanthanoids with special reference to oxidation state.
The chemistry of the actinoid elements is not so smooth as that of the Lanthanoids. Justify this statement by giving some examples from the oxidation state of these elements.
Name the members of the lanthanoid series which exhibit +4 oxidation state and those which exhibit +2 oxidation state. Try to correlate this type of behavior with the electronic configurations of these elements.
Account for the following:
Zn, Cd and Hg are soft metals.
Which trivalent ion has the maximum size in the Lanthanoid series, i.e., Lanthanum ion (La3+) to Luteium ion (Lu3+)?
(at. no. of Lanthanum = 57 and Lutetium = 71)
What is meant by Lanthanide contraction? Write the general electronic configuration of inner transition elements.
What is the action of the following on lanthanoids?
a. water
b. Sulphur, heat
c. nitrogen, heat
What is lanthanoid contraction?
Gadolinium belongs to 4f series. It’s atomic number is 64. Which of the following is the correct electronic configuration of gadolinium?
Which of the following lanthanoids show +2 oxidation state besides the characteristic oxidation state +3 of lanthanoids?
(i) \[\ce{Ce}\]
(ii) \[\ce{Eu}\]
(iii) \[\ce{Yb}\]
(iv) \[\ce{Ho}\]
Although +3 oxidation states is the characteristic oxidation state of lanthanoids but cerium shows +4 oxidation state also. Why?
Match the compounds/elements given in Column I with uses given in Column II.
| Column I (Compound/element) | Column II (Use) | |
| (i) | Lanthanoid oxide | (a) Production of iron alloy |
| (ii) | Lanthanoid | (b) Television screen |
| (iii) | Misch metal | (c) Petroleum cracking |
| (iv) | Magnesium based alloy is constituent of | (d) Lanthanoid metal + iron |
| (v) | Mixed oxides of lanthanoids are employed | (e) Bullets |
| (f) In X-ray screen |
Match the statements given in Column I with the oxidation states given in Column II.
| Column I | Column II | |
| (i) | Oxidation state of Mn in MnO2 is | (a) + 2 |
| (ii) | Most stable oxidation state of Mn is | (b) + 3 |
| (iii) | Most stable oxidation state of | (c) + 4 |
| Mn in oxides is | (d) + 5 | |
| (iv) | Characteristic oxidation state of lanthanoids is | (e) + 7 |
Match the property given in Column I with the element given in Column II.
| Column I (Property) | Column II (Element) | |
| (i) | Lanthanoid which shows +4 oxidation state |
(a) Pm |
| (ii) | Lanthanoid which can show +2 oxidation state |
(b) Ce |
| (iii) | Radioactive lanthanoid | (c) Lu |
| (iv) | Lanthanoid which has 4f7 electronic configuration in +3 oxidation state |
(d) Eu |
| (v) | Lanthanoid which has 4f14 electronic configuration in +3 oxidation state |
(e) Gd |
| (f) Dy |
On the basis of Lanthanoid contraction, explain the following:
Trends in the stability of oxo salts of lanthanoids from \[\ce{La}\] to \[\ce{Lu}\].
On the basis of Lanthanoid contraction, explain the following:
Stability of the complexes of lanthanoids.
The titanium (Z = 22) compound that does not exist is:-
In lanthanoid the last electron enters (n - 2)f subshell where n is equal to ______.
Zr (Z = 40) and Hf (Z = 72) have similar atomic and ionic radii because of ______.
The lathanide ion that would show colour is ______.
Write a note on lanthanoids.
Why is Mn2+ ion more stable than Fe2+ ion?
(Atomic numbers of Mn = 25 and Fe = 26)
The pair of lanthanoid ions which are diamagnetic is:
