Advertisements
Advertisements
Questions
What are the different oxidation states exhibited by the lanthanoids?
What are the oxidation states exhibited by lanthanoid elements?
What are the oxidation states exhibited by lanthanoids?
Advertisements
Solution 1
The members of group 3 of the periodic table are expected to have a uniform +3 oxidation state as a characteristic of the lanthanoids. The triply positive oxidation state is based on using the 6s2 electron and the lone 5d-electron or one of the f-electrons if no 5d-electron is present. The sum of the first three ionization enthalpies is relatively low, so that these elements are highly electropositive and readily form +3 ions. However, in an aqueous solution and in the solid state, cerium (Ce4+) can give quadripositive ions and samarium, europium and ytterbium (Sm2+, Eu2+ and Yb2+) can give bipositive ions. Other elements can give a +4 state in the solid state. Reduction of MX3 can give not only MX2 but also complex reduced species under special conditions.
The concept of +3 oxidation state for lanthanoids has become relatively rigid and other oxidation states are called ‘almost anomalous’. Such anomalous oxidation states of various lanthanoids are shown as follows:
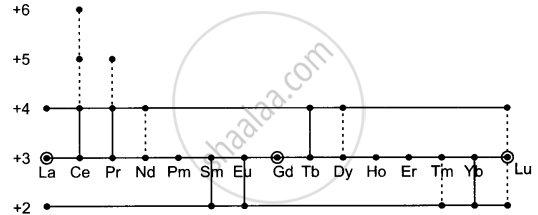
Different oxidation states are displayed by lanthanum and lanthanoids. Dotted lines show ambiguous or short-lasting valencies.
If we assume that special stability is associated with vacant, half-filled or full f-subshell, then the presence of +2 and +4 oxidation states can be reconciled with the electronic structures to a certain extent. Thus La, Gd and Lu form only tripositive ions because removing three electrons gives rise to the noble gas configuration in the La3+ ion. In Gd3 and Lu3+ ions, there is no removal of electrons from the stable configurations 4f7 and 4f14 respectively, because the lattice or hydration energies of M2+ or M+ ions will be smaller than those of M3+ ions or the additive lattice or hydration energies of salts of smaller M3+ ions.
The most stable bi- or quaternary ions are formed by elements that can do so by attaining f9, f7 and f14 configurations. Thus, cerium attains f0 configuration in the +4 oxidation state. Europium and ytterbium attain f7 and f14 configurations, respectively, in the +2 oxidation state. These facts seem to support the notion that the special stability of the f0, f7 and f14 configurations is important in determining the existence of oxidation states other than +3 for the lanthanoids, but this argument becomes less decisive when we see that samarium and thulium form M2+ ions, not M+ ions, having f6 and f13 configurations.
Also, praseodymium and neodymium form M4+ ions with f1 and f2 configurations but do not form penta- or hexavalent ions. There is no doubt that the Sm(II) and especially Tm(II), Pr(IV) and Nd(IV) states are very unstable, but the idea that merely approaching the f0, f7 or f14 configuration is conducive to stability even if no such configuration is attained is questionable. The existence of Nd2+(f4) is particularly conclusive evidence to believe that although the stability of the f0, f7 and f14 configuration may be one factor determining the stability of oxidation states, there are other thermodynamic and kinetic factors of equal or greater importance.
Solution 2
The lanthanoid elements (atomic numbers 57 to 71) primarily exhibit the +3 oxidation state, which is the most stable and common for all lanthanoids. However, some lanthanoids can also exhibit +2 and +4 oxidation states due to the variable involvement of 4f electrons.
APPEARS IN
RELATED QUESTIONS
Define lanthanoid contraction.
Write the different oxidation states of iron
Explain, why lanthanum (Z = 57) forms La3+ ion, while cerium (Z = 58) forms Ce4+ ion?
Explain effects of lanthanoid contraction.
What are the consequences of lanthanoid contraction?
Account for the following:
Zn, Cd and Hg are soft metals.
Which trivalent ion has the maximum size in the Lanthanoid series, i.e., Lanthanum ion (La3+) to Luteium ion (Lu3+)?
(at. no. of Lanthanum = 57 and Lutetium = 71)
Name an element of lanthanoid series which is well knwon to shown +4 oxidation state. Is it a strong oxidising agent or reducing agent?
What is lanthanoid contraction?
The f-block elements are known as ____________.
General electronic configuration of actinoids is `(n-2)f^(1-14)(n - 1)d^(0-2)ns^2`.Which of the following actinoids have one electron in 6d orbital?
(i) U (Atomic no. 92)
(ii) Np (Atomic no.93)
(iii) Pu (Atomic no. 94)
(iv) Am (Atomic no. 95)
On the basis of Lanthanoid contraction, explain the following:
Stability of the complexes of lanthanoids.
How would you account for the following:
There is a greater range of oxidation states among the actinoids than among the lanthanides.
Which of the following pairs has the same ionic size?
Write any two consequences of Lanthanoid Contraction.
Assertion: There is a continuous increase in size among Lanthanoids with an increase in atomic number.
Reason: Lanthanoids do not show Lanthanoid contraction.
Name an important alloy which contains some of the lanthanoid metals.
Name an important alloy which contains some of the lanthanoid metals. Mention its uses.
