Advertisements
Advertisements
Question
Elements A, B and C have atomic numbers 9, 20 and 10 respectively.
- State which one is
(1) a non-metal,
(2) a metal,
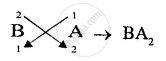
(3) chemically inert.[A,B,C] - Write down the formula of the compound formed by two of the above elements.[BA2]
Advertisements
Solution
- 9A 20B 10C
Atomic number of:
A = 9 = number of electrons = 2, 7
B = 20 = number of electons = 2, 8, 8, 2
C = 10 = number of electrons = 2, 8
(1) A non-metal = A
(2) A metal = B
(3) Chemically inert = C - Compound formed is A1−, B2+
APPEARS IN
RELATED QUESTIONS
Write true or false for the following statement
An atom on the whole has a positive charge.
Multiple Choice Questions
The sub atomic particle with negative charge is
Elements X, Y and Z have atomic numbers 6, 9 and 12 respectively. Which one:
- Forms an anion
- Forms a cation
- Has four electrons in its valence shell?
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Phosphorus – \[\ce{^31_15P}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 2, how many electrons does it have.
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Carbon [At. no. = 6, Mass no. = 12]
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Sodium [At. no. = 11, Mass no. = 23]
The atomic number of an element is 9, it has 10 neutrons. Find the element from the periodic table. What will be its mass number?
K and L shells can accommodate a maximum of 2 and 8 electrons respectively.
If M is the atomic mass and A is the mass number, then packing fraction is given by ______.
