Advertisements
Advertisements
Question
The atomic number and the mass number of an element is 26 and 56 respectively. Calculate the number of electrons, protons and neutrons in its atom. Draw the structure.
Advertisements
Solution
Given:
Atomic number A = 26
Mass number A = 56
No. of Protons P = ?
No. of Electrons = ?
No. of Neutrons N = ?
Name of the element is Iron (Fe)
No. of proton (P) = 26
Mass number A = n+p
56 = n = 26
n = 56-26
∴ No. of neutrons n = 30
In an element No. of protons and No. of electrons is equal.
∴No of electrons = no. of protons
= 26
Structure of Iron:
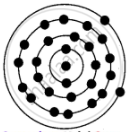
APPEARS IN
RELATED QUESTIONS
Which of the following are isoelectronic species i.e., those having the same number of electrons?
Na+, K+, Mg2+, Ca2+, S2–, Ar
The atomic number and the mass number of sodium are 11 and 23 respectively. What information is conveyed by this statement.
Fill in the following blanks in respect of an atom of an element
| No. of protons | No. of neutron | Mass number | Atomic number | No. of electrons | Symbol |
| 11 | 12 | ___________ | ___________ | ___________ | ________ |
The properties of the elements are a periodic function of their __________.
What is the significance of atomic numbers in the modern Periodic Table?
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Calcium – \[\ce{^40_20Ca}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
From the symbol \[\ce{^4_2He}\] for the element helium, write down the mass number and the atomic number of the element.
The particles that make up the atom are called ______ particles.
What is the relation between mass number & atomic number?
An element has 2 electrons in its N shell.
What is its atomic number?
