Advertisements
Advertisements
प्रश्न
The atomic number and the mass number of an element is 26 and 56 respectively. Calculate the number of electrons, protons and neutrons in its atom. Draw the structure.
Advertisements
उत्तर
Given:
Atomic number A = 26
Mass number A = 56
No. of Protons P = ?
No. of Electrons = ?
No. of Neutrons N = ?
Name of the element is Iron (Fe)
No. of proton (P) = 26
Mass number A = n+p
56 = n = 26
n = 56-26
∴ No. of neutrons n = 30
In an element No. of protons and No. of electrons is equal.
∴No of electrons = no. of protons
= 26
Structure of Iron:
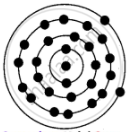
APPEARS IN
संबंधित प्रश्न
If number of electrons in an atom is 8 and number of protons is also 8, then
- What is the atomic number of the atom? and
- What is the charge on the atom?
What are the two main features of Rutherford’s atomic model?
WRITE SHORT ANSWER
Define the atomic mass number of an atom.
Fill in the Blank of the Following Statement :
An atom has atomic mass number 23 and atomic number 11. The atom has .................... electrons.
The mass number of an element is 23 and it contains 11 electrons. What is the number of protons and neutrons in it? What is the atomic number of the element?
From the symbol `""_12^24"Mg"`
, state the mass number, the atomic number and electronic configuration of magnesium.
Give reason
Actual atomic mass is greater than mass number.
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Are isotopes atoms of the same element or different elements.
Differentiate mass number from atomic number.
What are nucleons? How many nucleons are present in Phosphorous? Draw its structure.
