Advertisements
Advertisements
प्रश्न
The atomic number and the mass number of an element is 26 and 56 respectively. Calculate the number of electrons, protons and neutrons in its atom. Draw the structure.
Advertisements
उत्तर
Given:
Atomic number A = 26
Mass number A = 56
No. of Protons P = ?
No. of Electrons = ?
No. of Neutrons N = ?
Name of the element is Iron (Fe)
No. of proton (P) = 26
Mass number A = n+p
56 = n = 26
n = 56-26
∴ No. of neutrons n = 30
In an element No. of protons and No. of electrons is equal.
∴No of electrons = no. of protons
= 26
Structure of Iron:
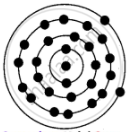
APPEARS IN
संबंधित प्रश्न
In Milikan’s experiment, static electric charge on the oil drops has been obtained by shining X-rays. If the static electric charge on the oil drop is - 1.282 × 10-18C, calculate the number of electrons present on it.
Fill in the blanks- The number of protons in the nucleus of an atom is called its ....................
Fill in the blank of the following statement :
The total number of protons and neutrons in the nucleus of an atom is called its _________.
Fill in the Blank of the Following Statement :
An atom has atomic mass number 23 and atomic number 11. The atom has .................... electrons.
The atom of an element X has 7 electrons in its M shell.
What is the atomic number of element X?
Match the atomic numbers 4,8,10,15 and 19 with the following:
Element with four shells
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 3, how many neutrons does it have.
Elements X, Y, Z have atomic numbers 6, 9 and 12 respectively. Which one:
- forms anion – negative ion;
- forms cation – positive ion;
- has 4 electrons in the outermost orbit. [Y, Z, X]
Elements A, B and C have atomic numbers 9, 20 and 10 respectively.
- State which one is
(1) a non-metal,
(2) a metal,
(3) chemically inert.[A,B,C] - Write down the formula of the compound formed by two of the above elements.[BA2]
What are nucleons? How many nucleons are present in Phosphorous? Draw its structure.
