Advertisements
Advertisements
प्रश्न
Elements A, B and C have atomic numbers 9, 20 and 10 respectively.
- State which one is
(1) a non-metal,
(2) a metal,
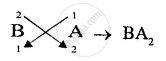
(3) chemically inert.[A,B,C] - Write down the formula of the compound formed by two of the above elements.[BA2]
Advertisements
उत्तर
- 9A 20B 10C
Atomic number of:
A = 9 = number of electrons = 2, 7
B = 20 = number of electons = 2, 8, 8, 2
C = 10 = number of electrons = 2, 8
(1) A non-metal = A
(2) A metal = B
(3) Chemically inert = C - Compound formed is A1−, B2+
APPEARS IN
संबंधित प्रश्न
Which of the following are isoelectronic species i.e., those having the same number of electrons?
Na+, K+, Mg2+, Ca2+, S2–, Ar
What are the atomic numbers of elements whose outermost electrons is represented by 3p5?
FILL IN THE BLANK
Rutherford won the Nobel Prize in 1908 for his investigations in the field of ......................
The number of electrons in the atom of an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of an atom of this element ?
The atomic number and mass number of sodium are 11 and 23 respectively. What information is conveyed by this statement?
Define the term of atomic number.
What is mass number of an atom?
In a neutral atom number of protons is equal to the number of neutrons.
If an atom has 6 electrons in its outermost shell, it is inert.
An element has 2 electrons in its N shell.
What is its atomic number?
