Advertisements
Advertisements
प्रश्न
Elements A, B and C have atomic numbers 9, 20 and 10 respectively.
- State which one is
(1) a non-metal,
(2) a metal,
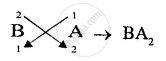
(3) chemically inert.[A,B,C] - Write down the formula of the compound formed by two of the above elements.[BA2]
Advertisements
उत्तर
- 9A 20B 10C
Atomic number of:
A = 9 = number of electrons = 2, 7
B = 20 = number of electons = 2, 8, 8, 2
C = 10 = number of electrons = 2, 8
(1) A non-metal = A
(2) A metal = B
(3) Chemically inert = C - Compound formed is A1−, B2+
APPEARS IN
संबंधित प्रश्न
Dalton said that ______ could not be divided.
Multiple Choice Questions
The sum of number of protons and number of neutrons present in the nucleus of an atom is called its
FILL IN THE BLANK
Atomic theory of matter was given by ..........................
From the symbol `""_15^31"P"`, state : atomic number of phosphorus
An atom of an element has two electrons in the M shell.
What is the (a) atomic number (b) number of protons in this element?
What is meant by the 'atomic number of an element'?
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Carbon [At. no. = 6, Mass no. = 12]
When an atom gives electrons from its outermost shell, it becomes ______ charged.
Mass No. 27 is for the element ______.
Match the following:
| Column A | Column B | ||
| 1. | Mass of proton | a. | +1 |
| 2. | Mass of electron | b. | -1 |
| 3. | Charge of electron | c. | 9.31 x 10-28g |
| 4. | Charge of proton | d. | 1.67 x 10-24g |
