Advertisements
Advertisements
Question
Each of the three states of matter has mass. Explain with the help simple experiments – that each state of matter has mass.
Advertisements
Solution
Matter i.e. solids, liquids, and Gases have mass explained by the following experiments.
(1) Solid
Experiment: A solid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the solid, hence all solids have mass.
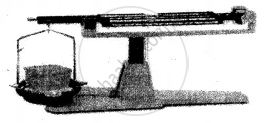
(2) Liquid:
Experiment: A liquid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the liquid, hence all liquids have mass.

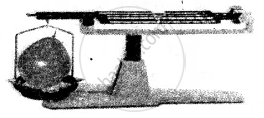
(3) Gas:
Experiment: An inflated balloon placed on one side of the scale causes it to tilt towards one side.
Conclusion: The scale tilts due to the mass of the gas, hence all gases have mass.
APPEARS IN
RELATED QUESTIONS
Give reasons:
A gas can fill the whole vessel in which it is enclosed.
Fill in the blank:
Water is a matter because it has mas and occupies....................
Fill in the blank:
Water boils at ............
A substance which can change its state
- wood
- oxygen
- paper
- cloth
Give reason:
A drop of ink added to water in a glass turns whole water blue.
If an object weighs 6 kg on earth. What will be its weight on moon ?
Name the three states of matter and define them.
Fill in the blanks:
Matter is defined as anything that occupies _______ and has _______
Select the correct answer from A, B, C, D & E for the statement given below:
The state of matter, where the inter-particle attraction between particles is maximum.
200 ml of water is poured into a bowl of 400 ml capacity. The volume of water will be ______.
