Advertisements
Advertisements
प्रश्न
Each of the three states of matter has mass. Explain with the help simple experiments – that each state of matter has mass.
Advertisements
उत्तर
Matter i.e. solids, liquids, and Gases have mass explained by the following experiments.
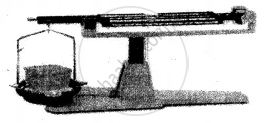
(1) Solid
Experiment: A solid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the solid, hence all solids have mass.
(2) Liquid:
Experiment: A liquid placed on one side of the scale, causes the scale to tilt towards one side.
Conclusion: The scale tilts due to the mass of the liquid, hence all liquids have mass.

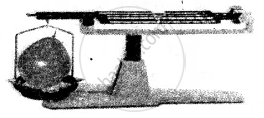
(3) Gas:
Experiment: An inflated balloon placed on one side of the scale causes it to tilt towards one side.
Conclusion: The scale tilts due to the mass of the gas, hence all gases have mass.
APPEARS IN
संबंधित प्रश्न
Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold, lemon water, smell of perfume.
Write your observation and conclusion for the following,
When a small stone is gently dipped into a glass filled with water.
Give reason of When a teaspoon of sugar is added to half a glass of water and stirred, the water level in the glass does not rise.
Fill in the blank
The physical state of a substance, which has a fixed volume but no fixed shape is _______
The state of matter which has no definite shape or volume is called
Even two or three crystals of potassium permanganate can impart colour to a very large volume of water. Which characteristic of particles of matter is illustrated by this observation?
Give reasons for the following:
Sugar can be distinguished from talcum powder using water.
On what basis are the three states classified of matter.
State in which of the three states of matter:
Are the atoms or particles far apart
Give a reason why solids and liquids co-exist at their melting points.
