Advertisements
Advertisements
Question
Define Hamburger’s phenomenon. Add a note on it.
Advertisements
Solution
Hamburger’s phenomenon or chloride shift refers to diffusion of chloride ions into the RBCs of to maintain the ionic balance between the RBCs and the plasma.
The rate of formation of carbonic acid inside the RBC is very high as compared to its formation in the plasma. Carbonic acid being unstable almost immediately dissociates into HCO3 − and H+ in the presence of the enzyme carbonic anhydrase (CA) leading to accumulation of large amount of HCO3 − inside the RBCs.
CO2 + H2O →CA ← H2CO3 →CA ← H+ + HCO3 − It thus, moves out of the RBCs causing an imbalance of the charge inside the RBCs. To maintain the ionic balance between the RBCs and the plasma, Cl- diffuses into the RBCs.
RELATED QUESTIONS
What is 'photophosphorylation'?
Describe non-cyclic photophosphorylation with schematic representation. Give its significance.
How is the process of respiration different from breathing?
Distinguish between inspiration and expiration.
After a long deep breath, we do not respire for some seconds due to ______.
Observe the figures given in Figures 10.3 (A) and (B) and answer the following.
In the figure label the arrows and indicate the direction of
- movement of air
- movement of the diaphragm
- movement of ribs
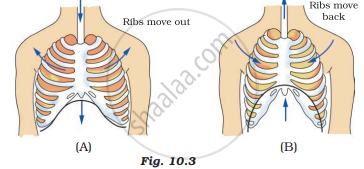
Describe the steps in breathing.
Complete the table:
| Partial pressure of gases |
Alveolar air | Pulmonary Capillaries |
| PP02 104 | mm Hg | ______ |
| PPC02 40 | mm Hg | ______ |
Interpret the given diagrams A and B. Enlist the changes occuring, during inspiration and expiration.

Percentage of carbon dioxide transported by bicarbonate ions, RBCs and plasma respectively is ______.
