Advertisements
Advertisements
प्रश्न
Define Hamburger’s phenomenon. Add a note on it.
Advertisements
उत्तर
Hamburger’s phenomenon or chloride shift refers to diffusion of chloride ions into the RBCs of to maintain the ionic balance between the RBCs and the plasma.
The rate of formation of carbonic acid inside the RBC is very high as compared to its formation in the plasma. Carbonic acid being unstable almost immediately dissociates into HCO3 − and H+ in the presence of the enzyme carbonic anhydrase (CA) leading to accumulation of large amount of HCO3 − inside the RBCs.
CO2 + H2O →CA ← H2CO3 →CA ← H+ + HCO3 − It thus, moves out of the RBCs causing an imbalance of the charge inside the RBCs. To maintain the ionic balance between the RBCs and the plasma, Cl- diffuses into the RBCs.
संबंधित प्रश्न
Very short answer question.
Why is a gas exchange very rapid at the alveolar level?
The volume of air that remains in the lungs after maximum respiration is ______.
With the help of labelled diagram explain the exchange of gases between alveolus and capillary.
______ nerve is located at the end of the eyes behind the retina.
Resistance in the airways is typically low. Why? Give two reasons.
During inspiration, the inter-costal muscles ____________.
Which of the following is formed in RBCs when CO2 combines with H2O in presence of carbonic anhydrase?
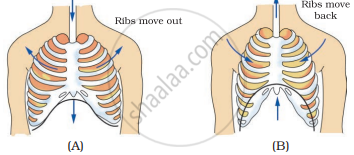
Observe the figures given in figure (A) and (B) and answer the following.
- Which of the figures A or B indicates the process of inhalation and which the process of exhalation?
- In the figure label the arrows and indicate the direction of
- movement of air
- movement of diaphragm
- movement of ribs
In breathing movements, air volume can be estimated by ______.
What is phosphorylation?
