Advertisements
Advertisements
Questions
Calculate the shortest wavelength of electromagnetic radiation present in Balmer series of hydrogen spectrum.
Calculate the shortest wavelength of Balmer series.
Advertisements
Solution
For shortest wavelength in Balmer series, nf = 2, ni = ∞
`1/lambda = R [1/n_f^2 - 1/n_i^2]`
`1/lambda = R(1/(2)^2 -0)`
`1/lambda = R/4`
∴ `lambda = 4/"R"`
∴ R = 1.097 × 107 m−1
` = 4/(1.097 xx 10^7) m`
` = 3.646 xx 10^-7 m`
= 3646 Å
= 364 nm
RELATED QUESTIONS
Name the electromagnetic radiations used for (a) water purification, and (b) eye surgery.
Name the subjective property of light related to its wavelength.
Give one use of ultraviolet radiation.
Moseley's Law for characteristic X-ray is √v = a(Z − b). Here,
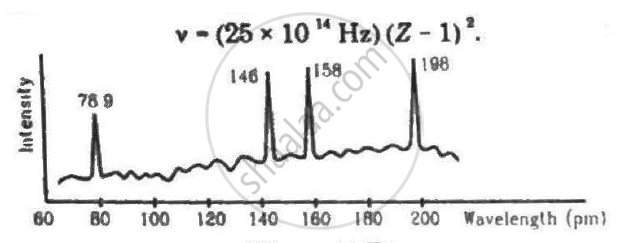
Continuous X-rays are made to strike a tissue paper soaked with polluted water. The incoming X-rays excite the atoms of the sample by knocking out the electrons from the inner shells. Characteristic X-rays are analysed and the intensity is plotted against the wavelength. Assuming that only Kα intensities are detected, list the elements present in the sample from the plot. Use Moseley's equation v − (25 × 1014Hz)(Z − 1)2.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the scientist who discovered Microwaves
Arrange the following electromagnetic waves in increasing order of their frequencies (i.e. begin with the lowest frequency):
Visible light, y rays, X rays, microwaves, radio waves, infrared radiations, and ultraviolet radiation.
One requires 11eV of energy to dissociate a carbon monoxide molecule into carbon and oxygen atoms. The minimum frequency of the appropriate electromagnetic radiation to achieve the dissociation lies in ______.
What is the wavelength range of electromagnetic radiation used in radio broadcast?
Name two electromagnetic waves of wavelength smaller than that of violet light.
