Advertisements
Advertisements
Question
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) |  |
| (II) |  |
| (III) |  |
Advertisements
Solution
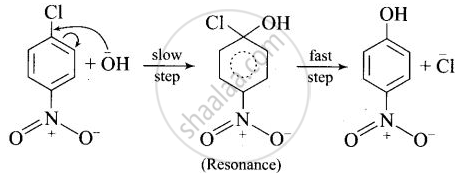
After the attachment of the nucleophile at the carbon carrying -Cl, the intermediate compound is stabilised due to resonance. Due to electron-withdrawing nature of-NO2, the nucleophile is easily attached to the benzene ring. Greater the number of -NO2 groups in the molecule, greater will be the ease with which the nucleophile will be attached. Hence, the order of reactivity is III > II > I.
APPEARS IN
RELATED QUESTIONS
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?

What are ambident nucleophiles? Explain with an example.
How will you bring about the following conversion?
Toluene to benzyl alcohol
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH(Br)CH2CH3 + NaOH ->[water]}\]
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
Halogenation of alkanes is ____________.
Most reactive halide towards SN1 reaction is ____________.
In the SN1 reaction, racemization takes place. It is due to:
Which of the following is an optically active compound?
Racemic compound has ____________.
An organic molecule necessarily shows optical activity if it ____________.
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Assertion: KCN reacts with methyl chloride to give methyl isocyanide.
Reason: CN– is an ambident nucleophile.
In which reaction mechanism carbocation is formed?
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with (i) NaOH (ii) dil. HNO3 (iii) AgNO3?
 |
 |
 |
 |
| I | II | III | IV |
The number of chiral carbons present in the molecule given below is ______.

Retention of configuration is observed in ______.
