Advertisements
Advertisements
Question
How will you represent first order reactions graphically.
Advertisements
Solution
i. The differential rate law for the first-order reaction A → P is 
The equation is of the form y = mx + c. A plot of rate versus [A]t is a straight line passing through the origin. The slope of straight line = k.
 |
| Variation of rate with [A] |
ii. The integrated rate law is
k = `2.303/t log_10 ["A"]_0/["A"]_"t"`
On rearrangement, the equation becomes
`(kt)/2.303 = log_10 ["A"]_0 - log_10 ["A"]_"t"`
Hence,
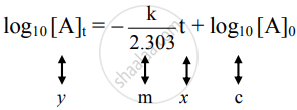
The equation is of the straight line. A graph of `log_10[A]_t` versus t yields a straight line with slope `-"k"/2.303` and y-axis intercepts as log10[A]0.
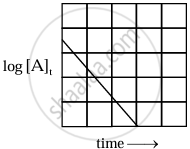 |
| Variation of `log_10 [A]_t` with time |
iii. Rearranging the integrated rate law equation, we get
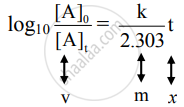
The equation has a straight-line form y = mx. Hence, the graph of `log_10 ([A]_0)/([A]_t)` versus t is a straight line passing through the origin.
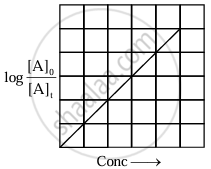 |
| Variation of `log_10 ([A]_0)/([A]_t)` with time |
APPEARS IN
RELATED QUESTIONS
What are pseudo-first-order reactions?
In a first-order reaction, the concentration of the reactant decreases from 20 mmol dm−3 to 8 mmol dm−3 in 38 minutes. What is the half-life of reaction?
Derive the integrated rate law for the zeroth order reaction.
Time required for 100% completion of a zero order reaction is _______.
Rate constant of a reaction is 3.6 × 10–3 s–1. The order of reaction is ______.
For first order reaction, the rate constant for the decomposition of N2O5 is 6 × 10–4 s –1. The half-life period for decomposition in seconds is ______.
The rate of catalysed reaction is large than the uncatalysed reaction as _______.
Write order of the following reaction:
\[\ce{2NH_{3(g)} -> N_{2(g)} + 3H_{2(g)}}\]
Write a mathematical expression for integrated rate law for zero-order reaction.
Explain pseudo first order reaction with a suitable example.
Write units of rate constants for:
- First-order reaction
- Zero-order reaction

This reaction follows first-order kinetics. The rate constant at particular temperature is 2.303 × 10−2 hour−1. The initial concentration of cyclopropane is 0.25 M. What will be the concentration of cyclopropane after 1806 minutes? (log 2 = 0.3010)
Assertion: rate of reaction doubles when the concentration of the reactant is doubles if it is a first-order reaction.
Reason: rate constant also doubles.
What is the value of rate constant of first order reaction, if it takes 15 minutes for consumption of 20% of reactants?
The following reactions follow zero order kinetics, EXCEPT ____________.
How long would it take to electroplate a spoon with 0.1 mol of silver (108 g/mol) at a constant current of 2.0 A using AgNO3?
For first order reaction the concentration of reactant decreases from 0.2 to 0.1 M in 100 minutes. What is the rate constant of the reaction?
A first order reaction has rate constant 1 × 10−2 s−1. What time will, it take for 20 g or reactant to reduce to 5 g?
A reaction that is of the first order with respect to reactant A has a rate constant 6 min−1. If we start with [A]0 = 0.5 mol dm−3, when would [A] reach the value 0.05 mol dm−3?
The time taken for 80% of a first order reaction to be complete is approximately ____________.
A first order reaction, A → B takes 100 minutes for it's 90% completion. What is the rate constant of reaction?
What is the unit of rate constant for the zero order reaction?
The integrated rate law is a direct relationship between time and ______
Calculate half-life of a first order reaction in minute if the rate constant is 1 × 10-3 s-1.
Half-life of a first order reaction is 30 minutes and initial concentration of reactant is 0.1 M.
What is half-life of the reaction if initial concentration of reactant is doubled?
Consider the following reaction.
\[\ce{SO2(g) + 1/2 O2(g) <=>[K1] SO3(g)}\]
\[\ce{2SO3(g)<=>[K2] 2SO2(g) + O2(g)}\]
What is the relation between K1 and K2?
A first order reaction takes 10 minute for 30% completion. Find rate constant of the reaction.
Calculate half life of reaction if rate constant of first order reaction is 0.0178 minute−1.
If the half-life of a first-order reaction is 10 minutes, find the time required to decrease the concentration of the reactant from 0.08 M to 0.02 M.
