Advertisements
Advertisements
Question
An element with the largest atomic radius among the following is ______.
Options
Carbon
Nitrogen
Lithium
Beryllium
Advertisements
Solution
An element with the largest atomic radius among the following is lithium.
Explanation:
- The second period contains carbon, nitrogen, lithium, and beryllium.
- Over a period, the atomic radius decreases from left to right.
- In the second period, lithium is on the far left.
- Thus, out of C, N, and Be, lithium has the biggest atomic radius.
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Give the trends in atomic size on moving across the period left to right.
Why is the size of sodium is greater than magnesium?
Atomic radius is expressed in the unit _______.
The size of an atom depends on the number of valence electrons.
Which of the following is the correct order of size?
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
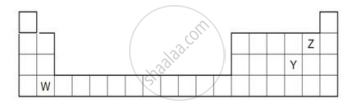
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
