Advertisements
Advertisements
Question
Why is the size of sodium is greater than magnesium?
Advertisements
Solution
Since atomic number of magnesium is more than sodium but the numbers of shells are same, the nuclear pull is more in case of Mg atom. Hence its size is smaller than sodium.
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
What do you understand by atomic size? State its unit?
Arrange the following in order of increasing radii:
CI- , CI
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
The following questions refer to the Periodic Table.
What happens to the atomic size of elements moving from top to bottom of a group?
The metals of Group 2 from top to bottom are Be, Mg, Ca, Sr and Ba. Which one of these elements will form ions most readily and why?
When an atom of iodine becomes an iodine ion (I–) the radius will ______
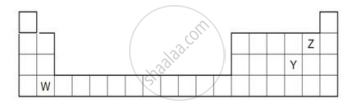
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following as per instruction given in the bracket.
Mg, Cl, Na, S, Si (decreasing order of atomic size)
