Advertisements
Advertisements
Question
Among NH3, H2O and HF, which would you expect to have highest magnitude of hydrogen bonding and why?
Advertisements
Solution
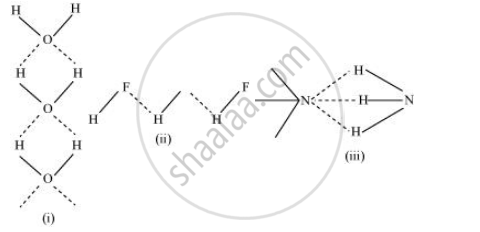
The extent of hydrogen bonding depends upon electronegativity and the number of hydrogen atoms available for bonding. Among nitrogen, fluorine, and oxygen, the increasing order of their electronegativities are N < O < F.
Hence, the expected order of the extent of hydrogen bonding is HF > H2O > NH3.
But, the actual order is H2O > HF > NH3.
Although fluorine is more electronegative than oxygen, the extent of hydrogen bonding is higher in water. There is a shortage of hydrogens in HF, whereas there are exactly the right numbers of hydrogens in water. As a result, only straight chain bonding takes place. On the other hand, oxygen forms a huge ring-like structure through its high ability of hydrogen bonding.
In case of ammonia, the extent of hydrogen bonding is limited because nitrogen has only one lone pair. Therefore, it cannot satisfy all hydrogens.
RELATED QUESTIONS
What properties of water make it useful as a solvent? What types of compound can it (i) dissolve, and (ii) hydrolyse?
Why is it said that-
There is no alternative to water for cleaning purposes.
State whether the following statement is true or false.
The substance in which a solute dissolves is called a solvent.
Why should a glass bottle completely filled with water never be kept in a freezer?
Define the following.
Freezing point
Which of the following equations depict the oxidising nature of \[\ce{H2O2}\]?
Which of the following equation depicts reducing nature of \[\ce{H2O2}\]?
If same mass of liquid water and a piece of ice is taken, then why is the density of ice less than that of liquid water?
Give reasons: Lakes freeze from top towards bottom.
Melting point, enthalpy of vapourisation and viscosity data of \[\ce{H2O}\] and \[\ce{D2O}\] is given below :
| \[\ce{H, O}\] | \[\ce{D2O}\] | |
| Melting point / K | 373.0 | 374.4 |
| Enthalpy of vapourisation at (373 K)/kJ mol–1 | 40.66 | 41.61 |
| Viscosity/centipoise | 0.8903 | 1.107 |
On the basis of this data explain in which of these liquids intermolecular forces are stronger?
Explain why \[\ce{HCl}\] is a gas and \[\ce{HF}\] is a liquid.
The freezing point of water ______ with an increase in pressure.
Freezing of water will cause an ______ is the volume.
What is the taste of distilled water & boiled water?
What are the physical properties of pure water?
Which of the following ion is responsible for the temporary hardness of water?
