Advertisements
Advertisements
प्रश्न
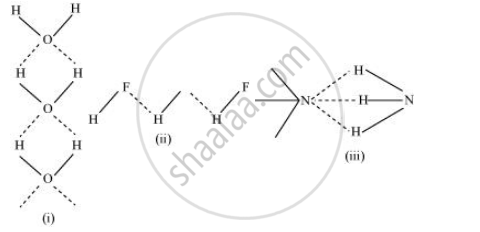
Among NH3, H2O and HF, which would you expect to have highest magnitude of hydrogen bonding and why?
Advertisements
उत्तर
The extent of hydrogen bonding depends upon electronegativity and the number of hydrogen atoms available for bonding. Among nitrogen, fluorine, and oxygen, the increasing order of their electronegativities are N < O < F.
Hence, the expected order of the extent of hydrogen bonding is HF > H2O > NH3.
But, the actual order is H2O > HF > NH3.
Although fluorine is more electronegative than oxygen, the extent of hydrogen bonding is higher in water. There is a shortage of hydrogens in HF, whereas there are exactly the right numbers of hydrogens in water. As a result, only straight chain bonding takes place. On the other hand, oxygen forms a huge ring-like structure through its high ability of hydrogen bonding.
In case of ammonia, the extent of hydrogen bonding is limited because nitrogen has only one lone pair. Therefore, it cannot satisfy all hydrogens.
संबंधित प्रश्न
Arrange the following:
CaH2, BeH2 and TiH2 in order of increasing electrical conductance.
Why is it said that-
Water is a universal solvent.
Why is it said that-
There is no alternative to water for cleaning purposes.
Explain the various properties of water.
Why should a glass bottle completely filled with water never be kept in a freezer?
Which of the following equations depict the oxidising nature of \[\ce{H2O2}\]?
Which of the following equation depicts reducing nature of \[\ce{H2O2}\]?
Give reasons: Lakes freeze from top towards bottom.
Give reasons: Ice floats on water.
Explain why \[\ce{HCl}\] is a gas and \[\ce{HF}\] is a liquid.
How will you account for 104.5° bond angle in water?
Pure water boils at ______ °C at one atmospheric pressure.
Freezing of water will cause an ______ is the volume.
One gram of water requires ______ of heat to raise its temperature by l°C.
Water is circulated around the car engine using the ______ pump and the heat is absorbed.
What are the physical properties of pure water?
Which of the following ion is responsible for the temporary hardness of water?
The water having more dissolved O2 is ______.
