Advertisements
Advertisements
Question
A particle moves in a closed orbit around the origin, due to a force which is directed towards the origin. The de Broglie wavelength of the particle varies cyclically between two values λ1, λ2 with λ1 > λ2. Which of the following statement are true?
- The particle could be moving in a circular orbit with origin as centre.
- The particle could be moving in an elliptic orbit with origin as its focus.
- When the de Broglie wavelength is λ1, the particle is nearer the origin than when its value is λ2.
- When the de Broglie wavelength is λ2, the particle is nearer the origin than when its value is λ1.
Options
b and d
a and c
b, c and d
a, c and d
Advertisements
Solution
b and d
Explanation:
According to the question, here given that the de-Broglie wavelength of the particle can be varying cyclically between two values λ1 and λ2, it is possible if particle is moving in an elliptical orbit with origin as its one focus.
As shown in the figure given alongside,
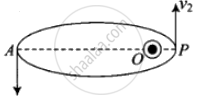
Let v1 and v2 be the speed of particle at A and B respectively and origin is at focus O. If λ1 and λ2 are the de-Broglie wavelengths associated with particle while moving at A and B respectively, then `λ_1 = h/(mv_1)`
And `λ_2 = h/(mv_2)`
∴ `λ_1/λ_2 = v_2/v_1`
Since `λ_1 > λ_2`
∴ `v_2 > v_1`
By the law of conservation of angular momentum, the particle moves faster when it is closer to focus.
From figure, we note that origin O is closed to P than A.
APPEARS IN
RELATED QUESTIONS
What is the de Broglie wavelength of a ball of mass 0.060 kg moving at a speed of 1.0 m/s?
For what kinetic energy of a neutron will the associated de Broglie wavelength be 1.40 × 10−10 m?
Find the de Broglie wavelength of a neutron, in thermal equilibrium with matter, having an average kinetic energy of `(3/2)` kT at 300 K.
Show that the wavelength of electromagnetic radiation is equal to the de Broglie wavelength of its quantum (photon).
A electron of mass me revolves around a nucleus of charge +Ze. Show that it behaves like a tiny magnetic dipole. Hence prove that the magnetic moment associated wit it is expressed as `vecμ =−e/(2 m_e)vecL `, where `vec L` is the orbital angular momentum of the electron. Give the significance of negative sign.
State any one phenomenon in which moving particles exhibit wave nature.
Describe briefly how the Davisson-Germer experiment demonstrated the wave nature of electrons.
When a light wave travels from air to glass ______.
What are matter waves?
Sodium and copper have work function 2.3 eV and 4.5 eV respectively. Then, the ratio of the wavelengths is nearest to ______.
The wavelength of the matter wave is dependent on ______.
An alpha particle is accelerated through a potential difference of 100 V. Calculate:
- The speed acquired by the alpha particle, and
- The de-Broglie wavelength is associated with it.
(Take mass of alpha particle = 6.4 × 10−27 kg)
Two particles move at a right angle to each other. Their de-Broglie wavelengths are λ1 and λ2 respectively. The particles suffer a perfectly inelastic collision. The de-Broglie wavelength λ, of the final particle, is given by ______.
The ratio of wavelengths of proton and deuteron accelerated by potential Vp and Vd is 1 : `sqrt2`. Then, the ratio of Vp to Vd will be ______.
For which of the following particles will it be most difficult to experimentally verify the de-Broglie relationship?
E, c and `v` represent the energy, velocity and frequency of a photon. Which of the following represents its wavelength?
Matter waves are ______.
The graph which shows the variation of `(1/lambda^2)` and its kinetic energy, E is (where λ is de Broglie wavelength of a free particle):
