Advertisements
Advertisements
प्रश्न
A particle moves in a closed orbit around the origin, due to a force which is directed towards the origin. The de Broglie wavelength of the particle varies cyclically between two values λ1, λ2 with λ1 > λ2. Which of the following statement are true?
- The particle could be moving in a circular orbit with origin as centre.
- The particle could be moving in an elliptic orbit with origin as its focus.
- When the de Broglie wavelength is λ1, the particle is nearer the origin than when its value is λ2.
- When the de Broglie wavelength is λ2, the particle is nearer the origin than when its value is λ1.
विकल्प
b and d
a and c
b, c and d
a, c and d
Advertisements
उत्तर
b and d
Explanation:
According to the question, here given that the de-Broglie wavelength of the particle can be varying cyclically between two values λ1 and λ2, it is possible if particle is moving in an elliptical orbit with origin as its one focus.
As shown in the figure given alongside,
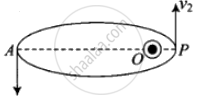
Let v1 and v2 be the speed of particle at A and B respectively and origin is at focus O. If λ1 and λ2 are the de-Broglie wavelengths associated with particle while moving at A and B respectively, then `λ_1 = h/(mv_1)`
And `λ_2 = h/(mv_2)`
∴ `λ_1/λ_2 = v_2/v_1`
Since `λ_1 > λ_2`
∴ `v_2 > v_1`
By the law of conservation of angular momentum, the particle moves faster when it is closer to focus.
From figure, we note that origin O is closed to P than A.
APPEARS IN
संबंधित प्रश्न
Calculate the de Broglie wavelength of the electrons accelerated through a potential difference of 56 V.
The wavelength of light from the spectral emission line of sodium is 589 nm. Find the kinetic energy at which
(a) an electron, and
(b) a neutron, would have the same de Broglie wavelength.
What is the de Broglie wavelength of a dust particle of mass 1.0 × 10−9 kg drifting with a speed of 2.2 m/s?
Find the de Broglie wavelength of a neutron, in thermal equilibrium with matter, having an average kinetic energy of `(3/2)` kT at 300 K.
Compute the typical de Broglie wavelength of an electron in a metal at 27°C and compare it with the mean separation between two electrons in a metal which is given to be about 2 × 10−10 m.
A electron of mass me revolves around a nucleus of charge +Ze. Show that it behaves like a tiny magnetic dipole. Hence prove that the magnetic moment associated wit it is expressed as `vecμ =−e/(2 m_e)vecL `, where `vec L` is the orbital angular momentum of the electron. Give the significance of negative sign.
Describe briefly how the Davisson-Germer experiment demonstrated the wave nature of electrons.
Why photoelectric effect cannot be explained on the basis of wave nature of light? Give reasons.
Show with the help of a labelled graph how their wavelength (λ) varies with their linear momentum (p).
70 cal of heat is required to raise the temperature of 2 moles of an ideal gas at constant pressure from 30°C to 35°C. The amount of heat required to raise the temperature of the gas through the same range at constant volume will be (assume R = 2 cal/mol-K).
An electromagnetic wave of wavelength ‘λ’ is incident on a photosensitive surface of negligible work function. If ‘m’ mass is of photoelectron emitted from the surface has de-Broglie wavelength λd, then ______
A particle is dropped from a height H. The de Broglie wavelength of the particle as a function of height is proportional to ______.
A proton, a neutron, an electron and an α-particle have same energy. Then their de Broglie wavelengths compare as ______.
An electron is moving with an initial velocity `v = v_0hati` and is in a magnetic field `B = B_0hatj`. Then it’s de Broglie wavelength ______.
An electron (mass m) with an initial velocity `v = v_0hati (v_0 > 0)` is in an electric field `E = - E_0hati `(E0 = constant > 0). It’s de Broglie wavelength at time t is given by ______.
An electron (mass m) with an initial velocity `v = v_0hati` is in an electric field `E = E_0hatj`. If λ0 = h/mv0, it’s de Broglie wavelength at time t is given by ______.
Two particles A1 sand A2 of masses m1, m2 (m1 > m2) have the same de Broglie wavelength. Then ______.
- their momenta are the same.
- their energies are the same.
- energy of A1 is less than the energy of A2.
- energy of A1 is more than the energy of A2.
The De-Broglie wavelength of electron in the third Bohr orbit of hydrogen is ______ × 10-11 m (given radius of first Bohr orbit is 5.3 × 10-11 m):
In a Frank-Hertz experiment, an electron of energy 5.6 eV passes through mercury vapour and emerges with an energy 0.7 eV. The minimum wavelength of photons emitted by mercury atoms is close to ______.
How will the de-Broglie wavelength associated with an electron be affected when the velocity of the electron decreases? Justify your answer.
