Advertisements
Advertisements
प्रश्न
A particle moves in a closed orbit around the origin, due to a force which is directed towards the origin. The de Broglie wavelength of the particle varies cyclically between two values λ1, λ2 with λ1 > λ2. Which of the following statement are true?
- The particle could be moving in a circular orbit with origin as centre.
- The particle could be moving in an elliptic orbit with origin as its focus.
- When the de Broglie wavelength is λ1, the particle is nearer the origin than when its value is λ2.
- When the de Broglie wavelength is λ2, the particle is nearer the origin than when its value is λ1.
पर्याय
b and d
a and c
b, c and d
a, c and d
Advertisements
उत्तर
b and d
Explanation:
According to the question, here given that the de-Broglie wavelength of the particle can be varying cyclically between two values λ1 and λ2, it is possible if particle is moving in an elliptical orbit with origin as its one focus.
As shown in the figure given alongside,
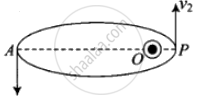
Let v1 and v2 be the speed of particle at A and B respectively and origin is at focus O. If λ1 and λ2 are the de-Broglie wavelengths associated with particle while moving at A and B respectively, then `λ_1 = h/(mv_1)`
And `λ_2 = h/(mv_2)`
∴ `λ_1/λ_2 = v_2/v_1`
Since `λ_1 > λ_2`
∴ `v_2 > v_1`
By the law of conservation of angular momentum, the particle moves faster when it is closer to focus.
From figure, we note that origin O is closed to P than A.
APPEARS IN
संबंधित प्रश्न
Describe the construction of photoelectric cell.
A proton and an α-particle have the same de-Broglie wavelength Determine the ratio of their speeds.
The wavelength of light from the spectral emission line of sodium is 589 nm. Find the kinetic energy at which
(a) an electron, and
(b) a neutron, would have the same de Broglie wavelength.
What is the de Broglie wavelength of a dust particle of mass 1.0 × 10−9 kg drifting with a speed of 2.2 m/s?
An electron and a photon each have a wavelength of 1.00 nm. Find
(a) their momenta,
(b) the energy of the photon, and
(c) the kinetic energy of electron.
Obtain the de Broglie wavelength of a neutron of kinetic energy 150 eV. As you have an electron beam of this energy is suitable for crystal diffraction experiments. Would a neutron beam of the same energy be equally suitable? Explain. (mn= 1.675 × 10−27 kg)
Obtain the de Broglie wavelength associated with thermal neutrons at room temperature (27°C). Hence explain why a fast neutron beam needs to be thermalised with the environment before it can be used for neutron diffraction experiments.
Compute the typical de Broglie wavelength of an electron in a metal at 27°C and compare it with the mean separation between two electrons in a metal which is given to be about 2 × 10−10 m.
The energy and momentum of an electron are related to the frequency and wavelength of the associated matter wave by the relations:
E = hv, p = `"h"/lambda`
But while the value of λ is physically significant, the value of v (and therefore, the value of the phase speed vλ) has no physical significance. Why?
Describe briefly how the Davisson-Germer experiment demonstrated the wave nature of electrons.
Sodium and copper have work function 2.3 eV and 4.5 eV respectively. Then, the ratio of the wavelengths is nearest to ______.
The de Broglie wavelength of a photon is twice the de Broglie wavelength of an electron. The speed of the electron is `v_e = c/100`. Then ______.
- `E_e/E_p = 10^-4`
- `E_e/E_p = 10^-2`
- `p_e/(m_ec) = 10^-2`
- `p_e/(m_ec) = 10^-4`
Assuming an electron is confined to a 1 nm wide region, find the uncertainty in momentum using Heisenberg Uncertainty principle (∆x∆p ≃ h). You can assume the uncertainty in position ∆x as 1 nm. Assuming p ≃ ∆p, find the energy of the electron in electron volts.
A particle A with a mass m A is moving with a velocity v and hits a particle B (mass mB) at rest (one dimensional motion). Find the change in the de Broglie wavelength of the particle A. Treat the collision as elastic.
A particle of mass 4M at rest disintegrates into two particles of mass M and 3M respectively having non zero velocities. The ratio of de-Broglie wavelength of particle of mass M to that of mass 3M will be:
An electron of mass me, and a proton of mass mp = 1836 me are moving with the same speed. The ratio of the de Broglie wavelength `lambda_"electron"/lambda_"proton"` will be:
How will the de-Broglie wavelength associated with an electron be affected when the velocity of the electron decreases? Justify your answer.
Matter waves are ______.
The graph which shows the variation of `(1/lambda^2)` and its kinetic energy, E is (where λ is de Broglie wavelength of a free particle):
