Advertisements
Advertisements
Question
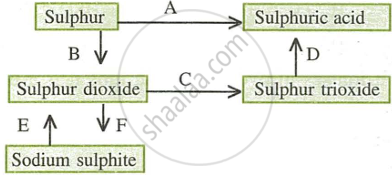
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
Advertisements
Solution
- Platinum or Vanadium pentoxide
- \[\ce{SO3 + H2SO4 -> H2S2O7}\]
- \[\ce{H2S2O7 + H2O -> 2H2SO4}\]
- The substance that will liberate sulphur dioxide in step E is dilute sulphuric acid (H2SO4).
- \[\ce{SO2 + 2NaOH -> Na2SO3 + H2O}\]
- \[\ce{Na2O + SO2 -> Na2SO3}\]
RELATED QUESTIONS
Quicklime is not used to dry HCl gas because _____.
Why is H2SO4 is not used as a drying agent for H2S?
Sulphuric acid can be used to prepare a number of gases in the laboratory. Write balanced equation for the reaction from which the folllowing gas are obtained, using dilute sulphuric acid as one of the reactant : Hydrogen
Sulphuric acid can be used to prepare a number of gases in the laboratory. Write balanced equation for the reaction from which the folllowing gas are obtained, using dilute sulphuric acid as one of the reactant : Sulphur dioxide
In this question, you required to supply the word (or words) that will make the sentence correct. Rewrite the copper statement.
Copper sulphate crystals are dehydrate by sulphuric acid.
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Lead sulphate from lead nitrate.
A, B, C and D summarize the properties of sulphuric acid depending on whether it is dilute or concentrated. Choose the property (A, B, C or D), depending on which is relevant to each of the preparations (i) to (ii).
A. Dilute acid (typical acid properties)
B. Non-volatile acid
C. Oxidizing agent
D. Dehydrating agent
(i) Preparation of hydrogen chloride.
(ii) Preparation of ethane from ethanol
(iii) Preparation of copper sulphate from copper oxide.
Give one equation to show the following property of sulphuric acid:
Dehydrating property
Identify the salts P and Q from the observation given below:
On performing the flame test salt P produces a lilac coloured flame and its solution gives a white precipitate with silver nitrate solution. Which is soluble in ammonium hydroxide solution.
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
Give a balanced chemical equation for the above reaction.
