Advertisements
Advertisements
Question
88Ra226 experiences three α - decay. Find the number of neutrons in the daughter element.
Advertisements
Solution
Given: Mass number of 88Ra226 = 226
Atomic number of 88Ra226 = 88
Mass number of α = 4
Atomic number of α = 2
After 3 α decay, Mass number of the daughter element will be,
= 226 − (3 × 4)
= 226 − 12
= 214
Atomic number of daughter element is,
= 88 − (3 × 2)
= 88 − 6
= 82
∴ Number of neutrons = 214 − 82
= 132
APPEARS IN
RELATED QUESTIONS
Name the unit in which the work function of a metal is expressed.
A radioactive source emits three types of radiations. Name them.
(i) Name the radiations which are charged.
(ii) Name the radiations which are most penetrating.
(iii) Name the radiations which travel with the speed of light.
(iv) Name the radiations which have the largest mass.
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
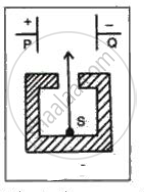
Why is the source S placed in a thick lead container?
Name two radioactive substances.
State three safety precautions that you would take while handling the radioactive substances.
Name a common device where a hot cathode ray tube is used.
Mention one important function of ear main parts:
State the principle on which the functioning of a nuclear reactor is based.
Two radioactive materials A and B have decay constants 25λ and 16λ respectively. If initially they have the same number of nuclei, then the ratio of the number of nuclei of B to that of A will be "e" after a time `1/("a"lambda)` The value of a ______.
Calculate the time interval between 33% decay and 67% decay if half-life of a substance is 20 minutes.
Assertion (A): Hydrogen atom consists of only one electron but its emission spectrum has may lines.
Reason (R): Only Lyman series is found in the absorption spectrum of hydrogen atom whereas in the emission spectrum, all the series are found.
