Advertisements
Advertisements
State the change in colour observed in following case mentioning the reason:
Silver chloride is exposed to sunlight.
Concept: undefined >> undefined
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
Concept: undefined >> undefined
Advertisements
When a green iron salt is heated strongly, its colour finally changes to brown and odour of burning sulphur is given out.
(a) Name the iron salt.
(b) Name the type of reaction that takes place during the heating of iron salt.
(c) Write a chemical equation for the reaction involved.
Concept: undefined >> undefined
Two metals X and Y form the salts XSO4 and Y2SO4, respectively. The solution of salt XSO4 is blue in colour whereas that of Y2SO4 is colourless. When barium chloride solution is added to XSO4 solution, then a white precipitate Z is formed alongwith a salt which turns the solution green. And when barium chloride solution is added to Y2SO4 solution, then the same white precipitate Z is formed alongwith colourless common salt solution.
(a) What could the metals X and Y be?
(b) Write the name and formula of salt XSO4.
(c) Write the name and formula of salt Y2SO4.
(d) What is the name and formula of white precipitate Z?
(e) Write the name and formula of the salt which turns the solution green in the first case.
Concept: undefined >> undefined
A metal salt MX when exposed to light splits up to form metal M and a gas X2. Metal M is used in making ornaments whereas gas X2 is used in making bleaching powder. The salt MX is itself used in black and white photography.
(a) What do you think metal M is?
(b) What could be gas X2?
(c) Name the metal salt MX.
(d) Name any two salt solutions which on mixing together can produce a precipitate of salt MX.
(e) What type of chemical reaction takes place when salt MX is exposed to light? Write the equation of the reaction?
Concept: undefined >> undefined
What does one mean by endothermic reaction? Give example.
Concept: undefined >> undefined
List in tabular form two differences between reflex action and walking.
Concept: undefined >> undefined
List in tabular form three distinguishing features between cerebrum and cerebellum.
Concept: undefined >> undefined
(a) Design an activity to demonstrate the decomposition reaction of lead nitrate.
(b) Draw labelled diagram of the experimental set-up. List two main observations.
(c) Write balanced chemical equation for the reaction stating the physical state of the reactant and the products.
Concept: undefined >> undefined
Two metallic spheres A and B kept on insulating stands are in contact with each other. A positively charged rod P is brought near the sphere A as shown in the figure. The two spheres are separated from each other, and the rod P is removed. What will be the nature of charges on spheres A and B?

Concept: undefined >> undefined
A metal sphere is kept on an insulting stands. A negatively charged rod is brought near it, then the sphere is earthed as shown. On removing the earthing, and taking the negatively charged rod away, what will be the nature of charge on the sphere? Give reason for your answer.
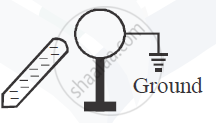
Concept: undefined >> undefined
Consider the sacle of voltmeter shown in the diagram and answer the following questions :

(a) What is the least count of the voltmeter?
(b) What is the reading shown by the voltmeter ?
(c) If the voltmeter is connected across a resistor of 20 `Omega` how much current is flowing through the resistor?
Concept: undefined >> undefined
A student wants to study a decomposition reaction by taking ferrous sulphate crystals. Write two precautions he must observe while performing the experiment.
Concept: undefined >> undefined
Answer the following question.
2 g of silver chloride is taken in a china dish and the china dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction.
Concept: undefined >> undefined
State the relation correlating the electric current flowing in a conductor and the voltage applied across it. Also, draw a graph to show this relationship.
Concept: undefined >> undefined
Classify the following reaction into different type:
\[\ce{2KClO3(s)->[\Delta] 2KCl(aq) + 3O2(g)}\]
Concept: undefined >> undefined
The following reaction is used for the preparation of oxygen gas in the laboratory:
\[\ce{2KClO3_{(s)} ->[Heat] 2KCl + 3O2_{(g)}}\]
Which of the following statement about the reaction is correct?
Concept: undefined >> undefined
Electrolysis of water is a decomposition reaction. The mole ratio of hydrogen and oxygen gases liberated during electrolysis of water is:
Concept: undefined >> undefined
