Please select a subject first
Advertisements
Advertisements
Convert the following:
Acetaldehyde to But-2-enal
Concept: Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
What happens when ethanal is treated with excess ethanol and acid?
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Write the main product in the following reaction:
\[\ce{CH3CH2CHO ->[Zn(Hg)/Conc. HCl]}\]
Concept: Chemical Reactions of Aldehydes and Ketones - Reduction
Write the main product in the following reaction:
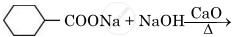
Concept: Uses of Carboxylic Acids
Which will undergo faster nucleophilic addition reaction?
Acetaldehyde or Propanone
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
What is the composition of Fehling's reagent?
Concept: Chemical Reactions of Aldehydes and Ketones - Oxidation
Which of the following is most reactive in nucleophilic addition reactions?
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
| The carbon-oxygen double bond is polarised in aldehydes and ketones due to higher electronegativity of oxygen relative to carbon. Therefore, they undergo nucleophilic addition reactions with a number of nucleophiles such as HCN, NaHSO3, alcohols, ammonia derivatives and Grignard reagents. Aldehydes are easily oxidised by mild oxidising agents as compared to ketones. The carbonyl group of carboxylic acid does not give reactions of aldehydes and ketones. Carboxylic acids are considerably more acidic than alcohols and most of simple phenols. |
Answer the following:
(a) Write the name of the product when an aldehyde reacts with excess alcohol in the presence of dry HCl. (1)
(b) Why carboxylic acid is a stronger acid than phenol? (1)
(c) (i) Arrange the following compounds in increasing order of their reactivity towards CH3MgBr: (1)
CH3CHO, \[\begin{array}{cc}
\ce{(CH3)3C-C-CH3}\\
\phantom{....}||\\
\phantom{....}\ce{O}
\end{array}\], \[\begin{array}{cc}
\ce{CH3-C-CH3}\\
||\\
\ce{O}
\end{array}\]
(ii) Write a chemical test to distinguish between propanal and propanone. (1)
OR
(c) Write the main product in the following: (2)
| (i) | 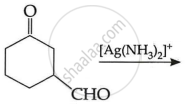 |
| (ii) |  |
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Why is the boiling point of aldehydes and ketones lower than that of corresponding carboxylic acids?
Concept: Physical Properties of Aldehydes and Ketones
An organic compound 'A' with molecular formula C5H8O2 is reduced to n-pentane with hydrazine followed by heating with NaOH and glycol. 'A' forms a dioxime with hydroxylamine and gives a positive iodoform and Tollen's test. Identify 'A' and give its reaction for iodoform and Tollen's test.
Concept: Chemical Reactions of Aldehydes and Ketones - Oxidation
Give a chemical test to distinguish between ethanal acid and ethanoic acid.
Concept: Physical Properties of Aldehydes and Ketones
An organic compound 'A' with the molecular formula C4H8O2 undergoes acid hydrolysis to form two compounds 'B' and 'C'. Oxidation of 'C' with acidified potassium permanganate also produces 'B'. Sodium salt of 'B' on heating with soda lime gives methane.
- Identify 'A', 'B' and 'C'.
- Out of 'B' and 'C', which will have higher boiling point? Give reason.
Concept: Chemical Reactions of Aldehydes and Ketones - Oxidation
Arrange the following: C2H5NH2, C2H5OH, (CH3)3N – in the increasing order of their boiling point
Concept: Physical Properties of Amines
Write the structures of A, B and C in the following:

Concept: Chemical Reaction of Diazonium Salts - Reactions Involving Displacement of Nitrogen
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Concept: Preparation of Amines
Write the structures of main products when aniline reacts with the following reagents :
Br2 water
Concept: Chemical Reactions of Amines - Electrophilic Substitution
Write the structures of main products when aniline reacts with the following reagents : HCl
Concept: Chemical Reactions of Amines - Basic Character of Amines
Write the structures of main products when aniline reacts with the following reagents :
(CH3CO)2O/pyridine
Concept: Chemical Reactions of Amines - Electrophilic Substitution
Give a simple chemical test to distinguish between the following pair of compounds :
(CH3)2NH and (CH3)3N
Concept: Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
Give the structures of A, B and C in the following reactions :

Concept: Preparation of Amines
