Advertisements
Advertisements
प्रश्न
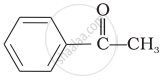
Which of the following compounds is most reactive towards nucleophilic addition reactions?
पर्याय
\[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\phantom{}\ce{CH3 - C - H}
\end{array}\]\[\begin{array}{cc}
\phantom{.}\ce{O}\\
\phantom{.}||\\
\phantom{}\ce{CH3 - C - CH3}
\end{array}\]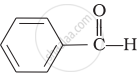
Advertisements
उत्तर
\[\begin{array}{cc}
\phantom{.....}\ce{O}\\
\phantom{.....}||\\
\phantom{}\ce{CH3 - C - H}
\end{array}\]
Explanation:
CH3CHO is most reactive towards nucleophilic addition reactions. Carbonyl compounds are polar with a positive charge on the carbon atom, which is attacked by nucleophiles. Two electron releasing alkyl groups in ketones make carbon less electron deficient than aldehydes. Benzene ring exhibits a +R effect, which thereby decreases the ease of nucleophilic addition reactions in benzaldehyde and acetophenone. Hence the reactivity order is:
\[\begin{array}{cc}
\phantom{...}\ce{H}\phantom{................}\ce{H}\phantom{.............}\ce{R}\phantom{..............}\\
\backslash\phantom{................}\backslash\phantom{...............}\backslash\phantom{.......}\\
\phantom{.......}{\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}} > \phantom{..}\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}} > \phantom{..}\overset{δ+}{\ce{C}} = \overset{δ-}{\ce{O}}}\phantom{}\\
/\phantom{................}/\phantom{...............}/\phantom{.......}\\
\phantom{...}\ce{H}\phantom{................}\ce{R}\phantom{.............}\ce{R}\phantom{..............}
\end{array}\]
