Advertisements
Advertisements
प्रश्न
The time required for 10% completion of a first order reaction at 298 K is equal to that required for its 25% completion at 308 K. If the value of A is 4 × 1010 s−1. Calculate k at 318 K and Ea.
Advertisements
उत्तर
t = `2.303/k_1 log [R]_0/(90/100 [R]_0)`
t = `2.303/k_2 log [R]_0/(75/100 [R]_0)`
t = `2.303/k_1 log 10/9`,
t = `2.303/k_2 log 4/3`
`2.303/k_1 log 10/9 = 2.303/k_2 log 4/3`
`=> k_2/k_1 = (log(4/3))/(log(10/9))`
= `log 1.333/log 1.111`
= `0.1249/0.0457`
= 2.733
`log k_2/k_1 = E_a/(2.303 R)((T_2 - T_1)/(T_1 T_2))`
⇒ log 2.733 = `E_a/(2.303 xx 8.314) ((308 - 298)/(298 xx 308))`
Ea = `(2.303 xx 8.314 xx 308 xx 298)/10 xx 0.4367`
= `(19.147 xx 308 xx 298)/10 xx 0.4367`
= 76.75 kJ mol−1
ln k = ln A `- E_a/(RT)`
log k = `log A - E_a/(2.303 RT)`
= `log (4 xx 10^10) - (76.75 xx 1000)/(2.303 xx 8.314 xx 318)`
= `10.6021 - 76750/6088.791`
= 10.6021 − 12.6051
= −2.003
k = Antilog (−2.003) = 9.93 × 10−3
Notes
The answer in the textbook is incorrect.
APPEARS IN
संबंधित प्रश्न
A first order reaction has a rate constant 1.15 × 10−3 s−1. How long will 5 g of this reactant take to reduce to 3 g?
In a pseudo first order hydrolysis of ester in water, the following results were obtained:
| t/s | 0 | 30 | 60 | 90 |
| [A]/mol L−1 | 0.55 | 0.31 | 0.17 | 0.085 |
Calculate the average rate of reaction between the time interval 30 to 60 seconds.
Following data are obtained for reaction :
N2O5 → 2NO2 + 1/2O2
| t/s | 0 | 300 | 600 |
| [N2O5]/mol L–1 | 1.6 × 10-2 | 0.8 × 10–2 | 0.4 × 10–2 |
1) Show that it follows first order reaction.
2) Calculate the half-life.
(Given log 2 = 0.3010, log 4 = 0.6021)
A first order reaction takes 20 minutes for 25% decomposition. Calculate the time when 75% of the reaction will be completed.
(Given : log = 2 = 0·3010, log 3 = 0·4771, log 4 = 0·6021)
Define order of reaction. How does order of a reaction differ from molecularity for a complex reaction?
Show that the time required for 99.9% completion of a first-order reaction is three times the time required for 90% completion.
Straight line graph for first order reaction is obtained between ____________.
Which of the following graphs is correct for a first order reaction?




The reaction X → product
Follow first order of kinetics. In 40 minutes the concentration of 'X' changes from 0.1 m to 0.025. M. The rate of reaction when concentration of X is 0.01 m is.
Observe the graph shown in figure and answer the following questions:
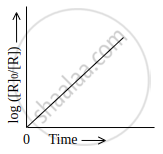
- What is the order of the reaction?
- What is the slope of the curve?
- Write the relationship between k and t1/2 (half life period).
The decomposition of formic acid on gold surface follows first-order kinetics. If the rate constant at 300 K is 1.0 × 10−3 s−1 and the activation energy Ea = 11.488 kJ mol−1, the rate constant at 200 K is ______ × 10−5 s−1. (Round off to the Nearest Integer)
(Given R = 8.314 J mol−1 K−1)
The slope in the plot of `log ["R"]_0/(["R"])` Vs. time for a first-order reaction is ______.
How will you represent first order reactions graphically?
What is the rate constant?
Slove: \[\ce{2NOBr -> 2NO_{2(g)} + Br_{2(g)}}\]
For the above reaction, the rate law is rate = k[NOBr]2. If the rate of reaction is 6.5 × 10−6 mol L−1 s−1 at 2 × 10−3 mol L−1 concentration of NOBr, calculate the rate constant k for the reaction.
Write the equation for integrated rate law for a first order reaction.
Show that `t_(1/2)= 0.693/k` for first reaction.
If the half-life (t1/2) for a first order reaction is 1 minute, then the time required for 99.9% completion of the reaction is closest to ______.
