Advertisements
Advertisements
प्रश्न
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
Advertisements
उत्तर
Given: k1 = 4.5 × 103 s−1
T1 = 273 + 10 = 283 K
k2 = 1.5 × 104 s−1
Ea = 60 kJ mol−1 = 6.0 × 104 J mol−1
T2 = ?
From Arrhenius equation, we obtain
`log k_2/k_1 = E_a/(2.303 R)((T_2 - T_1)/(T_1T_2))`
log `(1.5 xx 10^4)/(4.5 xx 10^3) = (60000)/(2.303 xx 8.314) ((T_2 - 283) /(283 xx T_2))`
or, log 3.333 = 3133.63`(T_2 - 283)/(283 xx T_2)`
or, `0.5228/3133.63 = (T_2 - 283)/(283 xx T_2)`
or, 0.0472T2 = T2 − 283
or, T2 = `283/0.9528`
= 297 K
= 297 − 273
= 24°C
Hence, k would be 1.5 × 104 s−1 at 24°C.
Notes
The answer in the textbook is incorrect.
APPEARS IN
संबंधित प्रश्न
The rate constant of a first order reaction increases from 4 × 10−2 to 8 × 10−2 when the temperature changes from 27°C to 37°C. Calculate the energy of activation (Ea). (log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
The rate constant for the first-order decomposition of H2O2 is given by the following equation:
`logk=14.2-(1.0xx10^4)/TK`
Calculate Ea for this reaction and rate constant k if its half-life period be 200 minutes.
(Given: R = 8.314 JK–1 mol–1)
The activation energy for the reaction \[\ce{2 HI_{(g)} -> H2_{(g)} + I2_{(g)}}\] is 209.5 kJ mol−1 at 581K. Calculate the fraction of molecules of reactants having energy equal to or greater than activation energy?
The rate constant for the decomposition of hydrocarbons is 2.418 × 10−5 s−1 at 546 K. If the energy of activation is 179.9 kJ/mol, what will be the value of pre-exponential factor?
The rate constant of a first order reaction are 0.58 S-1 at 313 K and 0.045 S-1 at 293 K. What is the energy of activation for the reaction?
Define activation energy.
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
Predict the main product of the following reactions: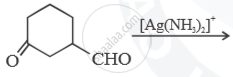
The rate of chemical reaction becomes double for every 10° rise in temperature because of ____________.
What happens to most probable kinetic energy and the energy of activation with increase in temperature?
For an endothermic reaction energy of activation is Ea and enthalpy of reaction ΔH (both of there in KJ moI–1) minimum value of Ea will be ______.
The rate constant for a reaction is 1.5 × 10–7 sec–1 at 50°C. What is the value of activation energy?
Explain how and why will the rate of reaction for a given reaction be affected when the temperature at which the reaction was taking place is decreased.
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below

The reaction must be:
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
It is generally observed that the rate of a chemical reaction becomes double with every 10°C rise in temperature. If the generalisation holds true for a reaction in the temperature range of 298 K to 308 K, what would be the value of activation energy (Ea) for the reaction?
Activation energy of any chemical reactions can be calculated if one knows the value of:
The rate of a reaction quadruples when temperature changes from 27°C to 57°C calculate the energy of activation.
Given: R = 8. 314 J K−1 mol−1, log 4 = 0.6021
