Advertisements
Advertisements
प्रश्न
Match the common names given in Column I with the IUPAC names given in Column II.
| Column I (Common names) |
Column II (IUPAC names) |
||
| (i) | Cinnamaldehyde | (a) | Pentanal |
| (ii) | Acetophenone | (b) | Prop-2-enal |
| (iii) | Valeraldehyde | (c) | 4-Methylpent-3-en-2-one |
| (iv) | Acrolein | (d) | 3-Phenylprop-2-enal |
| (v) | Mesityl oxide | (e) | 1-Phenylethanone |
जोड्या लावा/जोड्या जुळवा
Advertisements
उत्तर
| Column I (Common names) |
Column II (IUPAC names) |
||
| (i) | Cinnamaldehyde | (d) | 3-Phenylprop-2-enal |
| (ii) | Acetophenone | (e) | 1-Phenylethanone |
| (iii) | Valeraldehyde | (a) | Pentanal |
| (iv) | Acrolein | (b) | Prop-2-enal |
| (v) | Mesityl oxide | (c) | 4-Methylpent-3-en-2-one |
Explanation:
| (Common names) | Structure | (IUPAC names) |
| (i) Cinnamaldehyde | 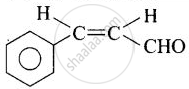 |
3-Phenylprop-2-enal |
| (ii) Acetophenone | 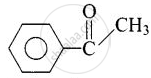 |
1-Phenylethanone |
| (iii) Valeraldehyde | 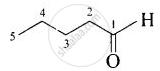 |
Pentanal |
| (iv) Acrolein | 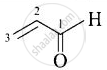 |
Prop-2-enal |
| (v) Mesityl oxide | 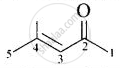 |
4-Methylpent-3-en-2- one |
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
