Advertisements
Advertisements
प्रश्न
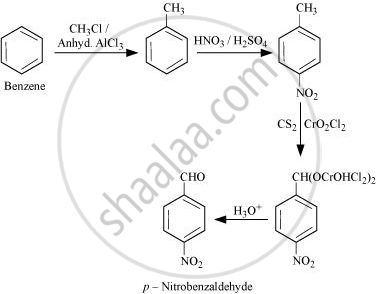
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzaldehyde
रासायनिक समीकरणे/रचना
Advertisements
उत्तर
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
पाठ 8: Aldehydes, Ketones and Carboxylic Acids - Exercises [पृष्ठ २५६]
