Advertisements
Advertisements
प्रश्न
Write the name of the reaction, structure and IUPAC name of the product formed when:
CH3CH2CN reacts with stannous chloride in the presence of hydrochloric acid, followed by hydrolysis.
Advertisements
उत्तर
Stephen reaction, CH3CH2CHO, Propanal.
APPEARS IN
संबंधित प्रश्न
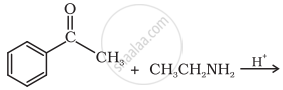
Predict the product of the following reaction:
When 0.4 g of acetic acid is dissolved in 40 g of benzene, the freezing point of the solution is lowered by 0.45 K. Calculate the degree of association of acetic acid. Acetic acid forms dimer when dissolved in benzene.
(Kf for benzene = 5.12 K kg mol−1, at. wt. C = 12, H = 1, O = 16)
Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
What is the name of the given reaction of preparation of aldehyde?
\[\ce{C3COCl ->[H2][Pd/BaSO4] CH3CHO + HCl}\]
The number of chiral carbon in glucose is:-
When 2 – hydroxyl benzoic acid distilled with zinc dust, it give
Aldehydes are the first oxidation products of ______.
The reaction of benzene with CO and HCl in the presence of anhydrous AlCl3 gives ______.
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
