Advertisements
Advertisements
प्रश्न
Explain the capillary action.
Advertisements
उत्तर
If one end of a capillary tube is dipped in a liquid which partially or completely wets the surface of the capillary, the level of liquid in the capillary rises. On the other hand, if the capillary tube is dipped in a liquid which does not wet its surface, the level of liquid in the capillary drops. The phenomenon of the rise or fall of a liquid inside a capillary tube when it is dipped in the liquid is called capillarity.


Consider the points A, B, C, and D such that
i) Point A is just above the convex surface and inside the capillary.
ii) Point B is just below the convex surface inside the capillary.
iii) Point C is just above the plane surface outside the capillary.
iv) Point D is just below the plane surface and outside the capillary, and below the point C.
Let PA, PB, PC, and PD be the pressure values at points A, B, C, and D, respectively. The pressure on the concave side is always greater than that on the convex side.
Capillary fall:
Consider a capillary tube dipped in a liquid which does not wet the surface. The shape of the liquid meniscus in the capillary is upper convex.
∴ PB > PA
As points A and C are at the same level, the pressure at both these points is the same, and it is the atmospheric pressure.
∴ PA = PC
Between the points C and D, the surface is plane.
∴ PC = PD = PA
∴ PB > PD.
But the points B and D are at the same horizontal level. Thus, in order to maintain the same pressure, the liquid in the capillary rushes out of the capillary. Because of this, there is a drop in the level of liquid inside the capillary, as shown.
Capillary rise:
Consider a capillary tube dipped in a liquid which wets the surface. The shape of the liquid meniscus in the capillary is concave.
∴ PA > PB
As points A and C are at the same level, the pressure at both these points is the same, and it is the atmospheric pressure.
∴ PA = PC
Between the points C and D, the surface is plane.
∴ PC = PD = PA
∴ PD > PB.
But the points B and D are at the same horizontal level. Thus, in order to maintain the same pressure, the liquid in the capillary rushes into the capillary. Because of this, there is a rise in the level of liquid outside the capillary, as shown.
APPEARS IN
संबंधित प्रश्न
Derive Laplace’s law for spherical membrane of bubble due to surface tension.
Derive an expression for excess pressure inside a drop of liquid.
Define the angle of contact.
In which of the following substances, surface tension increases with increase in temperature ?
- Copper
- Molten copper
- Iron
- Molten iron
Explain why The angle of contact of mercury with glass is obtuse, while that of water with glass is acute
What is the excess pressure inside a bubble of soap solution of radius 5.00 mm, given that the surface tension of soap solution at the temperature (20 °C) is 2.50 × 10–2 N m–1? If an air bubble of the same dimension were formed at depth of 40.0 cm inside a container containing the soap solution (of relative density 1.20), what would be the pressure inside the bubble? (1 atmospheric pressure is 1.01 × 105 Pa).
Mercury has an angle of contact equal to 140° with soda lime glass. A narrow tube of radius 1.00 mm made of this glass is dipped in a trough containing mercury. By what amount does the mercury dip down in the tube relative to the liquid surface outside? Surface tension of mercury at the temperature of the experiment is 0.465 N m–1. Density of mercury = 13.6 × 103 kg m–3
If water in one flask and castor oil in other are violently shaken and kept on a table, which will come to rest earlier?
A heavy mass is attached to a thin wire and is whirled in a vertical circle. The wire is most likely to break
If more air is pushed in a soap bubble, the pressure in it
A barometer is constructed with its tube having radius 1.0 mm. Assume that the surface of mercury in the tube is spherical in shape. If the atmospheric pressure is equal to 76 cm of mercury, what will be the height raised in the barometer tube? The contact angle of mercury with glass = 135° and surface tension of mercury = 0.465 N m−1. Density of mercury = 13600 kg m−3.
Find the surface energy of water kept in a cylindrical vessel of radius 6.0 cm. Surface tension of water = 0.075 J m−2.
Two large glass plates are placed vertically and parallel to each other inside a tank of water with separation between the plates equal to 1 mm. Find the rise of water in the space between the plates. Surface tension of water = 0.075 Nm−1.
Consider an ice cube of edge 1.0 cm kept in a gravity-free hall. Find the surface area of the water when the ice melts. Neglect the difference in densities of ice and water.
Find the force exerted by the water on a 2 m2 plane surface of a large stone placed at the bottom of a sea 500 m deep. Does the force depend on the orientation of the surface?
A metal piece of mass 160 g lies in equilibrium inside a glass of water. The piece touches the bottom of the glass at a small number of points. If the density of the metal is 8000 kg/m3, find the normal force exerted by the bottom of the glass on the metal piece.

A ferry boat has internal volume 1 m3 and weight 50 kg.(a) Neglecting the thickness of the wood, find the fraction of the volume of the boat immersed in water.(b) If a leak develops in the bottom and water starts coming in, what fraction of the boat's volume will be filled with water before water starts coming in from the sides?
A hollow spherical body of inner and outer radii 6 cm and 8 cm respectively floats half-submerged in water. Find the density of the material of the sphere.
How much amount of work is done in forming a soap bubble of radius r?
Calculate the rise of water inside a clean glass capillary tube of radius 0.1 mm, when immersed in water of surface tension 7 × 10-2 N/m. The angle of contact between water and glass is zero, the density of water = 1000 kg/m3, g = 9.8 m/s2.
A drop of mercury of radius 0.2 cm is broken into 8 droplets of the same size. Find the work done if the surface tension of mercury is 435.5 dyn/cm.
Insect moves over the surface of water because of ______.
The water droplets are spherical in free fall due to ______
Define surface tension.
Two soap bubbles have a radius in the ratio of 2:3. Compare the works done in blowing these bubbles.
Numerical Problem.
A stone weighs 500 N. Calculate the pressure exerted by it if it makes contact with a surface of area 25 cm2.
How does surface tension help a plant?
Mention the S.I unit and dimension of surface tension.
How is surface tension related to surface energy?
Define the angle of contact for a given pair of solid and liquid.
Distinguish between cohesive and adhesive forces.
What are the factors affecting the surface tension of a liquid?
Obtain an expression for the excess of pressure inside a
- liquid drop
- liquid bubble
- air bubble
What is capillarity?
Water rises in a capillary tube of radius r upto a height h. The mass of water in a capillary is m. The mass of water that will rise in a capillary of radius `"r"/4` will be ______.
The surface tension of the two liquids is respectively 20 and 60 dyne cm-1. The liquids drop from the ends of two tubes of the same radius. The ratio of the weights of the two drops is ______
A square frame of each side L is dipped in a soap solution and taken out. The force acting on the film formed is _____.
(T = surface tension of soap solution).
Water rises upto a height h in a capillary tube on the surface of the earth. The value of h will increase, if the experimental setup is kept in [g = acceleration due to gravity]
The upward force of 105 dyne due to surface tension is balanced by the force due to the weight of the water column and 'h' is the height of water in the capillary. The inner circumference of the capillary is ______.
(surface tension of water = 7 × 10-2 N/m)
Soap solution is used for cleaning dirty clothes because ______.
The wear and tear in the machine part is due to ______.
The length of a needle floating on water is 2 cm. The additional force due to surface tension required to pull the needle out of water will be (S.T. of water = 7.0 × 10−2 N/m).
The sap in trees, which consists mainly of water in summer, rises in a system of capillaries of radius r = 2.5 × 10–5 m. The surface tension of sap is T = 7.28 × 10–2 Nm–1 and the angle of contact is 0°. Does surface tension alone account for the supply of water to the top of all trees?
The sufrace tension and vapour pressure of water at 20°C is 7.28 × 10–2 Nm–1 and 2.33 × 103 Pa, respectively. What is the radius of the smallest spherical water droplet which can form without evaporating at 20°C?
We have three identical perfectly black plates. The temperatures of first and third plate is T and 3T. What is the temperature of second plate if system is in equilibrium?

A coaxial cylinder made of glass is immersed in liquid of surface tension ' S'. Radius of inner and outer surface of cylinder are R1 and R2 respectively. Height till which liquid will rise is (Density of liquid is p):
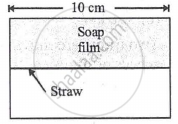
A soap film of surface tension 3 × 10-2 formed in a rectangular frame can support a straw as shown in Fig. If g = 10 ms-12, the mass of the straw is ______.
A liquid drop of density ρ is floating half immersed in a liquid of density d. The diameter of the liquid drop is ______.
(ρ > d, g = acceleration due to gravity, T = surface tension)
The surface tension of soap solution is 25 × 10-3 Nm-1. The excess of pressure inside a soap bubble of diameter 1 cm is ______.
Define angle of contact.
