Advertisements
Advertisements
प्रश्न
Draw neat and labelled diagram of Bessermer converter used in the extraction of copper.
Advertisements
उत्तर १
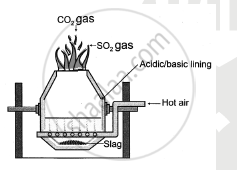
उत्तर २

APPEARS IN
संबंधित प्रश्न
Write the names and chemical formulae of any one ore of iron.
Write the reactions involved in the zone of reduction in blast furnace during extraction of iron.
Silica is added to roasted copper ore during smelting process to remove _______.
(A) ferrous sulphide
(B) ferrous oxide
(C) cuprous sulphide
(D) cuprous oxide
Write chemical formulae of the following ores :
(a) Calamine
(b) Haematite
(c) Magnetite
(d) Corundum
Out of C and CO, which is a better reducing agent at the lower temperature range in the blast furnace to extract iron from the oxide ore?
Mention names and formulae of two ores of alumunium.
What is the role of CO in the extraction of Iron?
Indicate the principle behind the method used for the refining of zinc.
Which form of the iron is the purest form of commercial iron?
Describe the roles of the following: SiO2 in the extraction of copper from copper matte
Which among the following minerals does NOT contain aluminium?
(a) Cryolite
(b) Siderite
(c) China clay
(d) Corundum
Explain the following methods to protect metals from corrosion:
Galvanization
Why does copper obtained in the extraction from copper pyrites have a blistered appearance?
How will you convert the following:
[Ag(CN)2]− to Ag
Give the uses of zinc.
Metals which occur in their native state in nature:
The most abundant metal on earth is:
When copper ore is mixed with silica, in a reverberatory furnace copper matte is produced. The copper matte contains ______.
In the extraction of aluminium by Hall-Heroult process, purified \[\ce{Al2O3}\] is mixed with \[\ce{CaF2}\] to:
(i) lower the melting point of \[\ce{Al2O3}\].
(ii) increase the conductivity of molten mixture.
(iii) reduce \[\ce{Al^{3+}}\] into \[\ce{Al(s)}\].
(iv) acts as catalyst.
Write the chemical reactions involved in the extraction of gold by cyanide process. Also give the role of zinc in the extraction.
Identify a mineral for the major source of boron from the following list?
The important ore of iron is:-
When compared to ΔG° for the formation of Al2O3 ΔG° for the formation of Cr2O3 is
Native silver metal forms a water solution complex with dilute solution of Na CN in the presence of
Match List I with List II.
| List - I | List - II | ||
| a. | Siderite | I. | FeCO3 |
| b. | Malachite | II. | CuCO3 · Cu(OH)2 |
| c. | Sphalerite | III. | ZnS |
| d. | Calamine | IV. | ZnCO3 |
Choose the correct answer from the option given below.
