Advertisements
Advertisements
प्रश्न
Draw neat and labelled diagram of Bessermer converter used in the extraction of copper.
Advertisements
उत्तर १
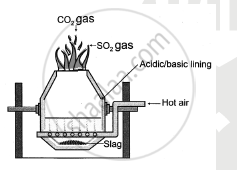
उत्तर २

APPEARS IN
संबंधित प्रश्न
Write the names and chemical formulae of any one ore of iron.
The process of leaching alumina, using sodium carbonate is called
- Bayer's process
- Decomposition
- Cyanide process
- Hall's process
Silica is added to roasted copper ore during smelting process to remove _______.
(A) ferrous sulphide
(B) ferrous oxide
(C) cuprous sulphide
(D) cuprous oxide
Out of C and CO, which is a better reducing agent at the lower temperature range in the blast furnace to extract iron from the oxide ore?
Purification of aluminium by electrolytic refining is carried out by
(a)Hoope process
(b)Hall process
(c)Baeyer process
(d)Serperck process
What is the role of CO in the extraction of Iron?
What is the role of coke in the extraction of iron from its oxides?
What is the role of silica in the extraction of copper?
Which of the ores mentioned can be concentrated by magnetic separation method?
Write the names and chemical formulae of any ‘two’ minerals of aluminium.
Which among the following minerals does NOT contain aluminium?
(a) Cryolite
(b) Siderite
(c) China clay
(d) Corundum
Explain the following methods to protect metals from corrosion:
Passivation
What is the role of the following compound-
SiO2 in the extraction of copper from copper pyrites?
A current liberates 0.50 g of hydrogen in 2 hours. The weight of copper (at. wt. = 63.5) deposited at the same time by the same current through copper sulphate solution is ______.
Describe the role of the cryolite in the extraction of aluminium from pure alumina.
How will you convert the following:
[Ag(CN)2]− to Ag
Metals which occur in their native state in nature:
Carbonate ore of iron is ____________.
Which of the following options are correct?
(i) Cast iron is obtained by remelting pig iron with scrap iron and coke using hot air blast.
(ii) In extraction of silver, silver is extracted as cationic complex.
(iii) Nickel is purified by zone refining.
(iv) Zr and Ti are purified by van Arkel method.
The main reactions occurring in blast furnace during extraction of iron from haematite are:
(i) \[\ce{Fe2O3 + 3CO -> 2Fe + 3CO2}\]
(ii) \[\ce{FeO + SiO2 -> FeSiO3}\]
(iii) \[\ce{Fe2O3 + 3C -> 2Fe + 3CO}\]
(iv) \[\ce{CaO + SiO2 -> CaSiO3}\]
Although carbon and hydrogen are better reducing agents but they are not used to reduce metallic oxides at high temperatures. Why?
Identify a mineral for the major source of boron from the following list?
The important ore of iron is:-
In the thermite process, the reducing agent is:
Extraction of zinc from zinc blende is achieved by:-
Which of the following is not an ore of magnesiuim?
Calamine and Malachite, respectively, are the ores of ______.
